Assign F or Z configuration to the following alkenes: (a) CH2 (b) CCH C=C C=C CH3CH2
Question:
Assign F or Z configuration to the following alkenes:
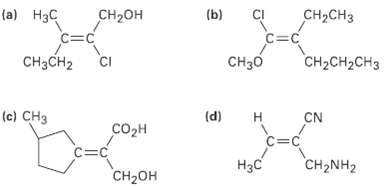
Transcribed Image Text:
(a) Нзс CH2он (b) CнаCHз C=C C=C CH3CH2 CI CH2CH2CH3 CH30 (e) CHз (d) н CN СOорн C=C C=C CH2NH2 НзС Cн-он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (14 reviews)
Low HC High CH2OH CC Low CH 0 Low High CH3CH High First consider the substituents on the right sid...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign R or S configuration to the following molecule, write the product you would expect from SN2 reaction with NaCN, and assign R or S configuration to the product (yellow-green =Cl):
-
An Assign E or Z configuration to each of the following alkenes: ( - C (a) H2 C=C C=C H3C ci OCH3 (c) (d) CH3O2C CH=CH2 NC CH C=C CH3CH2 CH2 HO2C CH-CH
-
Assign the configuration of the chirality center in the following compound:
-
Consider each of the following situations. Indicate whether (and why or why not) you think that the governement should account for the transactions and resources in an agency fund, a governmental...
-
Identify a personality trait you think would help a manager function as a contingency leader. Also, identify a trait you think would detract from a manager's ability to function as a contingency...
-
Reliable Insurance Company offers a term life insurance policy with a renewable annual premium. The first year premium is $795. Premiums increase by 4.1% each year. What will premiums be in the nth...
-
What are the two primary types of systems associated with system, product, or service development?
-
What are some of the reasons for the ARCs ethical dilemmas, and how can the organization guarantee that these problems will not recur in the future? The American Red Cross (ARC) is an independent...
-
4. On a frictionless pulley, a 4.0 and 6.0 kg mass are connected as shown on a ramp with a slope of 30 degrees. The coefficient of kinetic friction on the ramp is 0.18. a) Draw the FBDs of both...
-
Calculate the annual value of a specific purchase you make on a regular basis (for example, gasoline for your car). What would be the purchase value be over a 10 year period? What does this tell you...
-
Rank the following sets of substituents in order of Cahn-In-gold Prelog priorities: (a) CH3, 0H, H, C1 (b) CH3, CH2CH3, CH = CH2, CH2OH (c) CO2H, CH2OH, C = N, CH2NH2 (d) CH2CH3, C = CH, C = N,...
-
Assign stereochemistry (E or Z) to the double bond in the following compound, and convert the drawing into a skeletal structure (red0):
-
Which of the following has not been provided in IAS-7 but has been provided in AS-3? (a) Cash flow from operating activities (b) Cash flow from financing activities (c) Cash flow from investing...
-
You wish to test if two population means are equal to each other, against an alternative that they are different. You may assume equal variances. You have two samples n1-12 and n2-12. What is the...
-
For this assignment, assume that you were discussing what you learned in this module with a friend, and they were so impressed with your knowledge that they asked you to help to evaluate their...
-
Wildhorse Company incurred research and development costs of $97000 to develop a patent, and legal fees of $37000 to register the patent. The patent has a legal life of 20 years and a useful life of...
-
Surfside Incorporated produces surfboard wax. Total manufacturing costs are $360,000 when 80,000 containers of wax are produced. Of this amount, total variable costs are $120,000. What are the total...
-
Total fixed costs for Blue Dog Company are $62,000 Total costs, both fixed and variable are $210,000 if 90,000 units are produced. What would be the fixed cost per unit at 90,000 units?
-
How do weather fronts develop?
-
In what ways does a well-designed enterprise search software vary from popular search engines (e.g., Bing, DuckDuckGo, and Google)?
-
An 8.5-g ice cube is placed into 255 g of water. Calculate the temperature change in the water upon the complete melting of the ice. Assume that all of the energy required to melt the ice comes from...
-
For each of the Bronsted acid-base reactions shown in Fig. P3.26. label the conjugate acid-base pairs. Then give the curved-arrow notation for each reaction in the left-to-right direction. Figure...
-
For each of the Bronsted acid-base reactions shown in Fig. P3.26. label the conjugate acid-base pairs. Then give the curved-arrow notation for each reaction in the left-to-right direction. Figure...
-
Work Problem 3.27 for the reverse reactions in Fig. P3.27 Problem 3.27 (a) Classify each reaction step in the forward direction with one or more of the following terms: (1) A Lewis acid-base reaction...
-
Go into depth about the topic: Do women in the workplace still have to tolerate sexual harassment to advance or maintain their job positions? Why or why not? THE SCENARIO: You are human resource...
-
A pile group consists of nine friction piles in clay soil (see Figure 10-40). The diameter of each pile is 16 in., and the embedded length is 30 ft each. Center-to-center pile spacing is 4 ft. Soil...
-
Honest to Goodness Merchandising was registered as a Single Proprietorship by May Mendoza, the owner in the Department of Trade and Industry. Selected transactions for the month of July, 2019 are...

Study smarter with the SolutionInn App


