Both cis and trans alkenes can be formed from this compound y anti elimination. Draw a Newman
Question:
Both cis and trans alkenes can be formed from this compound y anti elimination. Draw a Newman projection of the conformation required to form each of these products and, on the basis of these projections predict which of these products would be formed in largeramounts?
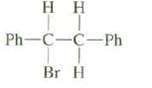
Transcribed Image Text:
нн Ph-C-C-Ph Br H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
The conformation that produces the tra...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw a Newman projection of the following compound, as viewed from the angle indicated. CI Br CI Br
-
In each case below, draw a Newman projection as viewed from the angle indicated: (a) (b) (c) (d) (e) (f) Observer CI Observer CI
-
Draw a Newman projection for each conformation the C2-C3 bond of isopentane, a compound containing a brached carbon chain. Show both staggered and eclipsed conformations. H,C CH CH, CH CHy isopentane
-
Following the 2017 General Election and change of government, the Labour-led coalition government established a Tax Working Group (TWG) to consider the overall structure, balance and fairness of the...
-
Cool Beat Sound (CBS) Ltd, which uses a job costing system, had two jobs in process at the start of the year: job number 64 ($84,000) and job number 65 ($53,500). The following information is...
-
Old Faithful is a famous geyser at Yellowstone National Park. From a sample with n = 32, the mean duration of Old Faithfuls eruptions is 3.32 minutes and the standard deviation is 1.09 minutes. Using...
-
What are the two primary types of systems associated with system, product, or service development?
-
Hunter, Folgers, and Tulip have been partners while sharing net income and loss in a 5:3:2 ratio. On January 31, the date Tulip retires from the partnership, the equities of the partners are Hunter,...
-
Read and evaluate the migrate with AWS article. Identify best practices you would include in your cloud-migration strategy. Also describes tools the AWS environment provides that may assist you in...
-
Q3. Consider the following frequency distribution about the age distribution of internet visitors to a travel site. Age Percent 18-24 11.30 25-34 19.10 35-44 23.64 45-54 23.48 55+ 22.48 a. Is this a...
-
What product would be expected from the elimination reaction of (1R, 2S)-1-bromo-1, 2-dipheylpropane using sodium ethoxide in ethanol as the solvent?
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
Victoria Ephanor manages a small product distribution company. Because the business is growing fast, Ephanor recognizes that it is time to manage the vast information pool to help guide the...
-
8) The figure shows the velocity-time graph of a particle which moves along a straight line from rest to rest in 30 seconds. The parts OA and AB are straight lines and the curve BC is an arc of the...
-
A and B have masses of 7 4 2 . 6 and 8 9 5 . 1 kg respectively. These masses are at the points ( 8 . 4 , 2 . 7 ) and ( 1 2 . 6 , 1 1 . 9 ) where all distances are measured in meters. What force does...
-
I f an ideal gas has a pressure of 6 . 4 7 0 1 0 5 Pa , a temperature of 8 1 . 0 0 c , and a volume of 0 . 0 5 6 9 m 3 , how many moles n of gas are in the sample?
-
What is the magntiude of the electric force felt in the below situations (in N)? A 10 uC charge is 2.0 m from a 30 uC charge. What is the magnitude of electric force that the 30 uC charge...
-
A galaxy known to be traveling away from the Earth at 0 . 7 0 0 percent of the speed of light is observed on Earth to have a strong peak in its spectrum at 6 3 3 . 1 nm . However, one small portion...
-
The process of removal of sprue and riser from casting is known as _____.
-
How much more interest will be earned if $5000 is invested for 6 years at 7% compounded continuously, instead of at 7% compounded quarterly?
-
Solve each inequality and graph its solution. 5(k + 6) > 15 -12 -10 do -6 T -2
-
Would you expect two diastereomers such as meso-2, 3-dibromobutanc and (2R, 3R)-dibromo butane to have identical or different IR spectra? Explain.
-
Propose structures for compounds that meet the following descriptions: (a) C5H8, with IR absorptions at 3300 and 2150 cm1 (b) C4H80, with a strong IR absorption at 3400 cm1 (c) C4H80, with a strong...
-
How could you use infrared spectroscopy to distinguish between the following pairs of isomers? (a) HC CCH2NH2 and CH3CH2C N (b) CH3COCH3 and CH3CH2CHO
-
Calculating Present Values Imprudential, Inc., has an unfunded pension liability of $645 million that must be paid in 25 years. To assess the value of the firm's stock, financial analysts want to...
-
Describe the operating activities of each company noting similarities and difference between COCA COLA & PEPSICO. Identify two economy wide factors and industry wide factors that could impact on the...
-
The trial balance for a company listed the following account balances at December 31, Year 1, the end of its fiscal year: cash, $36,000; accounts receivable. $31,000; Inventory, $45,000; equipment...

Study smarter with the SolutionInn App


