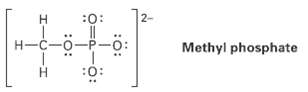
Organic phosphate groups occur commonly in biological molecules. Calculate formal charges on the four O atoms in
Question:
Organic phosphate groups occur commonly in biological molecules. Calculate formal charges on the four O atoms in the methyl phosphatedianion.
Transcribed Image Text:
2- :0: H-C-0-P-0: Methyl phosphate :0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
Formal charge FC of bonding electrons neering of v...View the full answer

Answered By

Joemar Canciller
I teach mathematics to students because I love to share what I have in this field.
I also want to see the students to love math and be fearless in this field.
I've been tutoring these past 2 years and I would like to continue what I've been doing.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c) H-C-N=N: H e) H H-=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
-
Calculate the formal charges on the atoms shown inred. (a) (CH3)2OBF3 (b) H2C-NEN: (c) H2C=N=N: (d) :=-: (e) H (f) - CH
-
Calculate formal charges for the non-hydrogen atoms in the following molecules: (a) Diazomethane, (b) Acetonitrile oxide, (c) Methylisocyanide, H2C=N=N: part a H3C-NEC: part b H3C-CEN-o: part c
-
Discuss the primary sources of nonverbal communication.
-
In general, the 45-day identification period and the 180-day exchange period for like-kind exchanges cannot be extended. Does this rule change if the like-kind property or the taxpayer involved in...
-
A "planet transit" is a rare celestial event in which a planet appears to cross in front of its star as seen from Earth. The planet transit causes a noticeable dip in the star's brightness, allowing...
-
What is the auditor's responsibility for subsequent events occurring after completion of field work but before issuance of his audit report.
-
A 17-year, $1,000 par value zero-coupon rate bond is to be issued to yield 7 percent. a. What should be the initial price of the bond? (Take the present value of $1,000 for 17 years at 7 percent,...
-
Walter Company has the following information for the month of March: Cash balance, March 1 Collections from customers Paid to suppliers Manufacturing overhead Direct labor Selling and administrative...
-
An information technology (IT) consulting firm specializing in healthcare solutions wants to study communication deficiencies in the health care industry. A random sample of 70 health care clinicians...
-
Nitromethane has the structure indicated. Explain why it must have formal charges on N andO. :0: Nitromethane :O:
-
Draw the indicated number of resonance forms for each of the following species: (a) The methyl phosphate anion, CH3OPO32- (3) (b) The nitrate anion, NO3- (3) (c) The allyl cation, H2C = CH ? CH2+ (2)...
-
The following account balances were included in the trial balance of J.R. Reid Corporation at June 30, 2008. The Retained Earnings account had a balance of $337,000 at July 1, 2007. There are 80,000...
-
When you blow air on the log in Problem 1, its temperature rises from 800 C to 950 C. How much thermal radiation does it emit now? P950C=i P800C
-
1. A magnetic field of 0.0200 T [up] is created in a region. (5 marks) a) Determine the magnitude and direction of the initial magnetic force on an electron initially moving at 5.00 x 106 m/s [N] in...
-
Define and explain the 3 types of financial costs: Fixed, Variable and Semi Variable costs. Give at least one example of each one.
-
An object moves in a circle of radius 22 m with its speed given by v = 3.6 + 1.5t2, with v in meters per second and t in seconds. At t = 3.5 s. Part A Find the tangential acceleration. Express your...
-
The human ear canal is about 2.8 cm long and can be regarded as a tube open at one end and closed at the eardrum. What is the fundamental frequency around which we would expect hearing to be most...
-
An economist wants to find the mean annual income for all adults in the United States. She knows that it is not practical to survey each member of the adult population, so she refers to an almanac...
-
What is the difference between the straight-line method of depreciation and the written down value method? Which method is more appropriate for reporting earnings?
-
The effect of pressure on the melting temperature of solids depends on the heat of fusion and the volume change on melting. The heat of fusion is always positive (that is, heat must be added to melt...
-
Provide IUPAC name for these alkenes: CH3 a) CHCHCHCHCH CH3 c) CHCHCHCHCH3 e) CHCH3 CH3 b) CHCHCHCHCH CHCH3 d) CHCHCHCHCCHCHCHCH T CH, CHCH CH, f)
-
Draw the structures of these compounds: (a) 4-Methylocatane (b) 2, 4-Dimethyl-5 propyl decane
-
What is wrong with these names? Provide the correct name for each. (a) 5, 5 Dimethyl-3-ethylhexane (b) 2-Dimethyl pentane
-
The wheels of a skateboard roll without slipping as it accelerates at 0.45 m/s down an 80-m-long hill. Part A If the skateboarder travels at 1.5 m/s at the top of the hill, what is the average...
-
A 2.30 g air-inflated balloon is given an excess negative charge, q = -3.50 10-8 C, by rubbing it with a blanket. It is found that a charged rod can be held above the balloon at a distance of d =...
-
A positive charge q = 2.60 C on a frictionless horizontal surface is attached to a spring of force constant k as in the figure shown below. When a charge of 42 = -8.350 C is placed 9.50 cm away from...

Study smarter with the SolutionInn App


