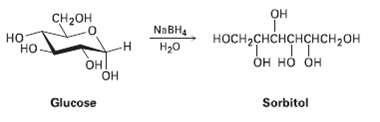
When glucose (Problem) is treated with NaBH4, reaction occurs to yield sorbitol, a polyalcohol commonly used as
Question:
When glucose (Problem) is treated with NaBH4, reaction occurs to yield sorbitol, a polyalcohol commonly used as a food additive. Show how this reductionoccurs.
Transcribed Image Text:
CH-он Он но NABHA носн-снснснснсн2он Он но он Нао н ОН но Он Sorbitol Glucose
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (14 reviews)
The free aldehyde form of glucose Problem ...View the full answer

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Sorbitol (D-glucitol) is used as a sugar substitute. Starting with maltose, suggest a synthesis of scorbitol.
-
Show how a Wittig reaction can be used to prepare each of the following compounds. In each case, also show how the Wittig reagent would be prepared: (a) (b)
-
When acetaldehyde is treated with aqueous acid, an aldol reaction can occur. In other words, aldol reactions can also occur in acidic conditions, although the intermediate is different than the...
-
Conduct an internal and external analysis of yourself in the current business industry. examples: you are an accounting major so your environment is the cpa job market. you are a finance major so...
-
1. What advantages and disadvantages to telecommuting is Angela experiencing? 2. What advantages and disadvantages to managing a telecommuter is Ralph experiencing?
-
Harris Company manufactures a single product. Costs for the year 2001 for output levels of 1,000 and 2,000 units are given in Table P8.4. At each level of output, compute the following: (a) Total...
-
Starting with Eq. (4.1-10), show that if \(\Delta v \ll \bar{v}\) and \(r \ll 2 c / \Delta v\) for all \(P_{1}\), then \[ \mathbf{u}\left(P_{0}, t ight) \approx \iint_{\Sigma} \frac{e^{j 2 \pi(r /...
-
Delta Company, a U.S. MNC, is contemplating making a foreign capital expenditure in South Africa. The initial cost of the project is ZAR10,000. The annual cash flows over the five-year economic life...
-
A 1200 kg truck traveling at 45.0 mph drives on a banked curve angled at 22.0 degrees. The radius of curvature for the banked curve is 55.0 m. Determine the amount of working done by the force of...
-
Here is the Situation of Black Elk Medical center (BEMC), which is a quality-driven acute care hospital organization that includes three medical facilities that treat both inpatients and outpatients....
-
When crystals of pure ?-glucose are dissolved in water, isomerization slowly occurs to produce ?-glucose. Propose a mechanism for the isomerization. CH2 CH2 - - B-Glucose a-Glucose
-
Give IUPAC names for the followingcompounds: (a) Br (b) (c) CH CH3CH2CHCH2CH2CH3 CHH-CH2Co CHCH2C (f) O2- (d) CH CN (e) CHCCH2CHH "Co2 CH2CH2COH
-
Rowen Company has accounts receivable of $241,000 at September 30, 2021. An analysis of the accounts shows the following: Month of Sale ..................... Balance September...
-
The spot price of oil is $80 per barrel and the cost of storing a barrel of oil for one year is $3, payable at the end of the year. The risk-free interest rate is 5% per annum, continuously...
-
Assume that an investment in the S&P 5 0 0 index gives an average return of 1 1 % . The short - term US government bond provides a risk - free return of 3 . 5 % . a . If you aim to earn an expected...
-
How do you prioritize features and attributes when designing a new product or service? What steps should be taken to ensure that the design process aligns with the organization's overall strategy and...
-
Suppose you are a British venture capitalist holding a major stake in an e-commerce start-up in Silicon Valley. As a British resident, you are concerned with the pound value of your U.S. equity...
-
B&H Co. expects its EBIT to be $50,000 every year forever. The firm can borrow at 6% interest rate B&H currently has no debt, and its cost of equity is 8%. The tax rate is 0%. Required 1.What is the...
-
Consider the Weil [1650] model introduced in Sect. 9.5. By applying Jensen's inequality, prove Proposition 9.15. Data From Proposition 9.15 Proposition 9.15 In the context of the above model (see...
-
Extend Algorithms 3.4 and 3.5 to include as output the first and second derivatives of the spline at the nodes.
-
Consider the phase diagram shown here. Identify the states present at points a through g. Pressure (not to scale) Pc- a. e 0Q g b G 14 Pl I Ic Temperature (not to scale)
-
Without consulting tables, select the stronger acid from each of the following pairs: (a) 4-Methylphenol and 4-fluorophenol (b) 4-Methylphenol and 4-nitrophenol (c) 4-Nitrophenol and 3-nitrophenol...
-
What products would be obtained from each of the following acid-base reactions? (a) Sodium ethoxide in ethanol + phenol (b) Phenol + aqueous sodium hydroxide (c) Sodium phenoxide + aqueous...
-
Describe a simple chemical test that could be used to distinguish between members of each of the following pairs of compounds: (a) 4-Chlorophenol and 4-chloro-1-methylbenzene (b) 4-Methylphenol and...
-
Athens Gas Station has figured out the weekly demand distribution for their gas sales. Each gallon of gas sold at the pump results in a profit of 10 cents/gallon and any lost sales results in a cost...
-
1. The maximum pressure an eardrum can withstand without rupturing is Pmax = 3.0102 Pa. Assume a sound is made at that pressure. If the density of air is, p=1.20 kg/m, and the speed of sound is 340...
-
52. A wave is sent down a string with a linear mass density of 1.600 x 10-4 kg/m. The wavefunction that describes this wave is: y(x,t) = (0.021 m)*sin{(2.00 rad/m)*x + (30.0 rad/s)*t} What is the...

Study smarter with the SolutionInn App


