Explain which of these compounds has a faster rate of E2elimination: CH CH3 CI 'CI
Question:
Explain which of these compounds has a faster rate of E2elimination:
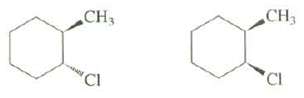
Transcribed Image Text:
CH CH3 CI 'CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
The cisisomer has a faster rate of E2 elimination because its more stable chair conformat...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain which of these compounds has the faster rate of reaction withBr2: Ph Ph - c=CH, or .
-
Which of these compounds would have faster rate of SN1 reaction? a) b) c) d) e) CI ta Ph Ph -CI or Br or Br or or -Cl or + CH3 Ph -Cl Br Br ta -CI
-
Which of these compounds would have a faster rate of SN2 reaction? a) b) c) d) e) CI CI Br or or Br or Br or or CI CI Br Br
-
Explain in detail the role of data analytics in healthcare.
-
Describe the two main steps involved in process costing.
-
VIP-MD is a health maintenance organization (HMO) located in North Carolina. Unlike the traditional fee-for-service model that determines the payment according to the actual services used or costs...
-
Use the implicit finite difference method to solve the heat conduction problem on the unit square: \[\begin{aligned}& \frac{\partial^{2} u}{\partial x^{2}}=\frac{\partial u}{\partial t} \\& u(x, 0)=x...
-
Selected financial data for Stanley Black & Decker, Inc. and Snap-On Tools for 2009 are presented here (in millions). Instructions(a) For each company, compute the following ratios.(1) Current...
-
Q10) There is a 48.84% probability of an average economy and a 51.16% probability of an above average economy. You invest 24.55% of your money in Stock S and 75.45% of your money in Stock T. In an...
-
Mr B aged 52 years, has earned rupees 75,00,000 out of his business. His ex-wife gifted him a car worth rupees 8 lakh. He spent a total of rupees 20 lakh during a family trip. He won a lottery of 16...
-
Explain why the reaction of the cis-isomer of this compound with potassium tert-but oxide in tert-butanol is about 500 times faster than that of thetrans-isomer. Br C(CH3)3
-
Frequently, several different routes can potentially be used to synthesize a desired compound. For example, the following two routes can be envisioned for the preparation of cyclopentyl methyl ether....
-
Several years ago, Stanfords restaurant in Lake Oswego was written up for how well it used HR to keep good employees. What do customers at sites such as...
-
Derive the expression for combined gas law.
-
Discuss about entropy and avaliable energy?
-
What strategies or programs could Meda utilize to improve employee well-being?
-
Discuss about entropy and Clausius inequality.
-
Define open, closed and isolated systems. Classify each with example.
-
Phase-distribution measurements were made for the PEG 3400 - dextran T40 - water system at \(4{ }^{\circ} \mathrm{C}\) by Diamond and Hsu (1989). The following table gives the equilibrium...
-
Which of the ocean zones shown would be home to each of the following organisms: lobster, coral, mussel, porpoise, and dragonfish? For those organisms you identify as living in the pelagic...
-
Evaluate each using the values given. z + x - (+3); use x = 1, and = = 4
-
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, ordiastereotopic: (c) (b) (a) - HC- - H3C FCH2
-
The following compounds all show a single line in their 1H NMR spectra. List them in expected order of increasing chemical shift: CH4, CH2C12, cyclohexane, CH3COCH3, H2C = CH2, benzene
-
Predict the splitting pattern for each kind of hydrogen in the following molecules: (a) (CH3)3CH (b) CH3CH2CO2CH3 (c) trans-2-Butene
-
Excerpts from Andre Company's December 31, 2024 and 2023, financial statements are presented below: Accounts receivable Inventory Net sales Cost of goods sold Total assets Net income Total...
-
The York City Hospital has just acquired new equipment. The equipment cost $ 4 , 2 5 0 , 0 0 0 , and the organization spent $ 1 3 5 , 0 0 0 on upgrading the physical plant to the new equipment will...
-
Calculate the following showing all the necessary steps: Note: CPP rate to be used in the calculation is 5.95%. Note: Don't forget to deduct pay period exemption\ \ Tanya earns $25.00 per hour. This...

Study smarter with the SolutionInn App


