Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, ordiastereotopic: (c) (b) (a) - HC- -
Question:
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, ordiastereotopic:
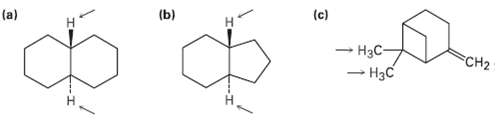
Transcribed Image Text:
(c) (b) (a) - HạC- - H3C FCH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
a homotopic ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, ordiastereotopic: (c) (a) (b) - C=C Hc CH (d) (e) (f) CH CH Br .
-
Determine whether the indicated sets of complex vectors are linearly independent or dependent. 1-i 2i 1- 5i 1 .11 2 230 20-2 +20 13 - + -- 212
-
Identify the indicated protons in the following molecules as unrelated, homotopic, enantiotopic, ordiastereotopic: (a) (b) Cysteine
-
1. Identify the function of the underlined noun/s in each item. 1. On rainy mornings, is your bus usually late? 2. On the desk were the red pencils. 3. Volcanoes and earthquakes are destructive...
-
What is the property rights theory of the firm? Is this theory consistent with the theories of vertical integration described in Chapter 3?
-
Determine Vo and Io in the circuit of Fig. 10.80 using mesh analysis. j4 4-30 A
-
A household goods company decides to produce a faucet mount water filter for removing organic and metallic contaminants from tap water. The filter is made up of activated carbon and ionexchange...
-
An investor purchased a one-acre lot on the outskirts of a city for $9000 cash. Each year he paid $80 of property taxes. At the end of 4 years, he sold the lot. After deducting his selling expenses,...
-
Pistol Pete's Mustache Mania recently reported net income of $5.2 million and depreciation of $800,000. What is was net cash flow? Assume it has no amortization expense.
-
A group of Hope College statistics students wanted to see if there was an association between students major and the time (in seconds) it takes them to complete a small Sudoku-like puzzle. They...
-
How many types of nonequivalent protons are present in each of the followingmolecules? (c) (b) CH3CH2CH20CH3 (a) H3C CH3 Naphthalene (e) (d) C=CH2 CO2CH2CH3 Ethyl acrylate Styrene
-
The following compounds all show a single line in their 1H NMR spectra. List them in expected order of increasing chemical shift: CH4, CH2C12, cyclohexane, CH3COCH3, H2C = CH2, benzene
-
If true, cite appropriate facts or theorems. If false, explain why or give a counterexample that shows why the statement is not true in every case. (T/F) If A is m x n and the linear transformation x...
-
Show whether or not the following formulae are dimensionally consistent? (a) h = 9(gt - 2vt) 8(v-gt) gt gt 2v 2va (b) t = (c) t = y V v sin 0+ sin2 0+2gy 9 (d) K = (kAr-mgAr sin a) cos a where k has...
-
[5 points] (d) A modem has a baud rate of 2400 symbols/sec. What would be the bit rate for this modem if its phase-amplitude constellation is as follows: [2 points] (e) A voice grade modem has a...
-
Z Incorporation acquired significant influence over Y Company by purchasing 1 0 0 percent of the common stock of the Y Incorporation for $ 1 0 0 , 0 0 0 ; Y earns income of $ 5 0 , 0 0 0 and pays...
-
Suppose the following balance sheet for Winbnb after second round of venture financing. Second Stage Market Value Balance Sheet ($mil) Assets Cash from new Equity Fixed Assets Liabilities and Equity...
-
Sanchez Co . sold for $ 1 2 , 0 0 0 inventory that had cost $ 8 , 0 0 0 . Freight terms for the sale were FOB destination and payment terms were 1 / 1 0 , n / 3 0 . Sanchez records sales transactions...
-
Find all possible values for each expression. \((\sqrt{3}-j)^{1 / 4}\)
-
As you rewrite these sentences, replace the cliches and buzzwords with plain language (if you don't recognize any of these terms, you can find definitions online): a. Being a jack-of-all-trades, Dave...
-
You can purchase hydrochloric acid in a concentrated form that is 37.0% HCl by mass and that has a density of 1.20 g/mL. Describe exactly how to prepare 2.85 L of 0.500 M HCl from the concentrated...
-
Determine the ratios of the peak areas in the following spectra. Then use this information, together with the chemical shifts, to pair up the compounds with their spectra. Assign the peaks in each...
-
Draw the NMR spectra you would expect for the following compounds. (a) (CH3)2CH-O-CH(CH3)2 (b) (c) Ph-CH(CH3)2 (d) (e) CI-CH2-CHC_O_CH3 - CH2COOCH CH CH2 COOCH2CH3
-
(a) Assign protons to the peaks in the NMR spectrum of 4, 4-dimethylcyclohex-2-en-1 one in Figure 13-30(a). Explain the splitting that gives the triplets at 1.8 and 2.4. (b) Assign protons to the...
-
You placed $6,599 in a savings account today that earns an annual interest rate of 3 percent compounded annually. How much you will have in this account at the end of 2 years?
-
You're planning a trip to France. The current exchange rate is 1.21 dollars per euro. If you want to get 3,000, how many dollars do you have to pay? If you want to exchange $3,000, how many euros...
-
The exchange rate between euros and dollars is currently 0.83 euros per dollar. Inflation is expected to be 1% in Europe and 2% in the US. If relative purchasing power parity holds, what is the...

Study smarter with the SolutionInn App


