Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm
Question:
Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm?1 in its IR spectrum and B has a peak at 1715cm?1, the mass spectra of A and B are as follows, show the structures of A and B.
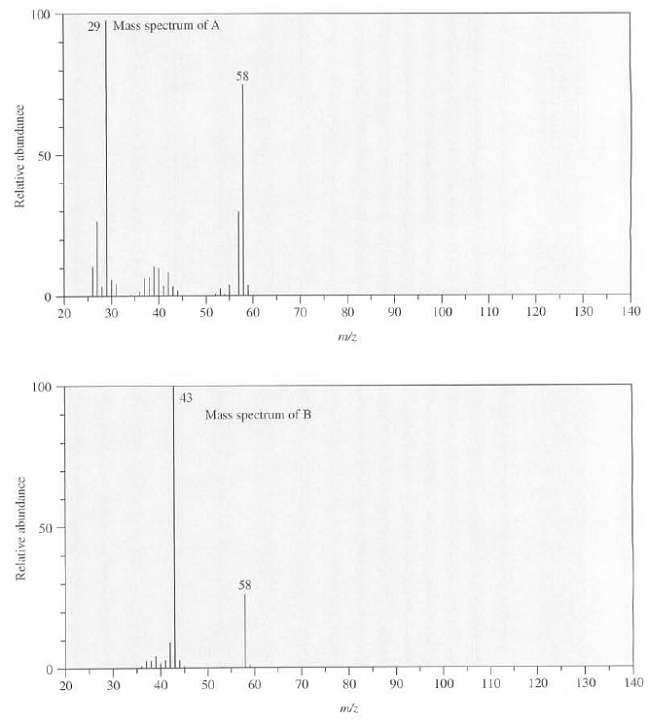
Transcribed Image Text:
100 29 | Mass spectrum of A 58 50 140 70 80 90 100 110 120 130 20 30 40 50 60 100 43 Mass spectrum of B 50 58 140 120 130 70 80 90 100 110 30 40 50 60 20 Relative abundance Relative abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The DU for both isomers is 1 Based on their PR spectra this must result from a carbonyl group ...View the full answer

Answered By

Gabriela Rosalía Castro
I have worked with very different types of students, from little kids to bussines men and women. I have thaught at universities, schools, but mostly in private sessions for specialized purpuses. Sometimes I tutored kids that needed help with their classes at school, some others were high school or college students that needed to prepare for an exam to study abroud. Currently I'm teaching bussiness English for people in bussiness positions that want to improve their skills, and preparing and ex-student to pass a standarized test to study in the UK.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra. Compound A: 1.3 ppm (3H, triplet); 3.6 ppm (2H, quartet); 4.1 ppm (2H,...
-
Compounds A and B are isomers of molecular formula C10H14. Identify each one on the basis of the 13C NMR spectra presented in Figure 13.41.
-
Compounds A, B, and C are isomers with the formula C5H11Br. Their broadband proton-decoupled 13C NMR spectra are given in Fig. 9.32. Information from the DEPT 13C NMR spectra is given near each peak....
-
At Blossom Company, events and transactions during 2020 included the following. The tax rate for all items is 20%. (1) Depreciation for 2018 was found to be understated by $148000. (2) A strike by...
-
How does administrative responsibility contribute to the attainment of public interest?
-
Let P(x) = anxn + an1xn1 + + a1x + a0 be a polynomial, and let x0 be given. Construct an algorithm to evaluate P(x0) using nested multiplication.
-
For a specially orthotropic, transversely isotropic material the "plane strain bulk modulus," \(K_{23}\), is an engineering constant that is defined by the stress conditions...
-
At the beginning of 2007 Ace Company had the following portfolio of investments in available-for-sale securities (common stock): During 2007 the following transactions occurred: May 3 Purchased C...
-
What role does phase equilibrium play in the design and optimization of liquid-liquid extraction processes, and how can mathematical modeling be used to predict extraction efficiency ?
-
Kohler Clothiers manufactures women's business suits. The company uses a standard cost accounting system. In March 2012, 15,700 suits were made. The following standard and actual cost data applied to...
-
The mass spectra of 3-ethyl-2-pentanone and 4-methyl-2-pentanone are as follows. Explain which spectrum goes with which compound, what is the structure of the ion responsible for the peak at m/z 43...
-
Compounds C and D are isomers with the formula C9H12, in addition to other absorption peaks, both compounds show a peak near 7.25 ? (area 5) in their 1H-NMR spectra. Their mass spectra are as follow,...
-
What factors affect the future movements in the value of the euro against the dollar?
-
Discuss the advantages of preparing a cash budget and the challenges a management accountant faces in the process of forecasting cash inflows and outflows. (10 marks) A Government Institute has...
-
What are the original points of differentiation of Facebook? Give an opinion about the outcome of the film The Social Network, including impressions of Zuckerberg, Parker, the Winklevoss twins.
-
Acme, Inc. instructed its bank, AJM Bank, to pay $500,000 to TreeTop, Inc. TreeTop, Inc. was also a customer of AJM Bank. AJM Bank executed the payment order by crediting Treetop's account with...
-
Transforming Healthcare , there is a discussion about how many doctors and nurses found the most difficult change to be the standardization of work. Dr. Jacobs describes how there can be six...
-
If you were opening a new slow-food restaurant in your town, how would you position it? Discuss the considerations that would go into this decision.
-
List three advantages of using pea plants as an experimental organism.
-
What kind of rays are X-rays?
-
Three economists at the Federal Reserve Bank of St. Louis Andrew Levin, Fabio Nattaluci, and Jeremy Piger have attempted to measure the effects of a short-lived increase in actual inflation on...
-
Write structures for all compounds with molecular formula C4H6O that would not be expected to exhibit infrared absorption in the 3200-3550-cm-1 and 1620-1780-cm-1 regions.
-
Add curved arrows to the following reactions to indicate the flow of electrons for all of the bond-forming and bond-breaking steps. (a) (b) H.
-
Most carboxylic acids dissolve in aqueous solutions of sodium bicarbonate (NaHCO3) because, as carboxylate salts, they are more polar. Write curved arrows showing the reaction between a generic...
-
x-8x+15 A) Let f(x) = = x2+2x-15 Calculate lim f(x) x 3 2x225x75 B) Let f(x) = x-18x45 Calculate lim f(x) x 15
-
If a dance instructor prices her lessons at $60 per student, she will have three students. If she prices her lessons at $50 per student, she will have four students. How much marginal revenue will...
-
What is the difference between objectives, strategies, and goals? How do these things improve our planning process?

Study smarter with the SolutionInn App


