Question: Explain how mass spectrometry could be used to distinguish between thesecompounds: a) and b) and c) and
Explain how mass spectrometry could be used to distinguish between thesecompounds:
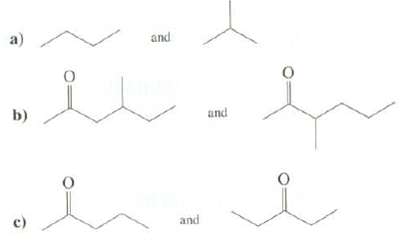
a) and b) and c) and
Step by Step Solution
3.31 Rating (178 Votes )
There are 3 Steps involved in it
a The major fragment In the mass spectrum of butane is an ethyl cation at mz ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-S (113).docx
120 KBs Word File


