Predict the major fragments and their m/z that would appear in the mass spectra of these compounds:
Question:
Predict the major fragments and their m/z that would appear in the mass spectra of these compounds:
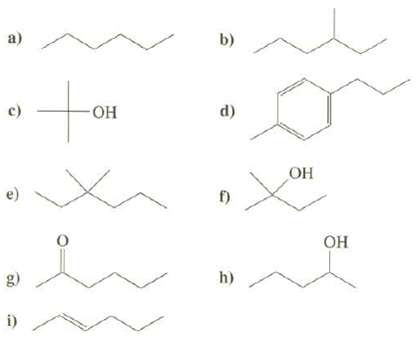
Transcribed Image Text:
a) b) c) -Он d) ОН г) Он h) g) i)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (18 reviews)

Answered By

Anthony Ngatia
I have three academic degrees i.e bachelors degree in Education(English & Literature),bachelors degree in business administration(entrepreneurship option),and masters degree in business administration(strategic management) in addition to a diploma in business management.I have spent much of my life in the academia where I have taught at high school,middle level colleges level and at university level.I have been an active academic essays writer since 2011 where I have worked with some of the most reputable essay companies based in Europe and in the US.I have over the years perfected my academic writing skills as a result of tackling numerous different assignments.I do not plagiarize and I maintain competitive quality in all the assignments that I handle.I am driven by strong work ethics and a firm conviction that I should "Do Unto others as I would Like them to do to me".
4.80+
76+ Reviews
152+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the 1H.NMR spectra of these compounds include the approximate chemical shift, multiplicity, and integral for each type ofhydrogen. CI b) CH;CHCH; ) C,CH,H c) CH,CH,OCH,CH3 CH2CH2NO2 f)...
-
What fragments might you expect in the mass spectra of the followingcompounds? (c) (b) "
-
The following are typical disclosures that would appear in the notes accompanying financial statements. For each of the items listed, indicate where the disclosure would likely appeareither in (A)...
-
A soil has a bulk density of 135 lb/ft 3 and a dry density of 120 lb/ft 3 , and the specific gravity of the soil particles is 2.75. Determine (a) moisture content, (b) degree of saturation, (c) void...
-
Watson manufactures and sells appliances. Intro develops and manufactures computer technology. Trenton operates general merchandise retail stores. Selected data for these companies appear in the...
-
Calculate the availability of the water at the initial and final states of Problem 8.70, and the irreversibility of the process.
-
What is a system property?
-
Preparing a Trial Balance Paige Consultants, Inc., provides marketing research for clients in the retail industry. The company had the following unadjusted balances at September 30, 2012: Required:...
-
How does capitalism fit in with modern business life and how does it impact ethical dilemmas that you have seen or heard of in the workplace? What are two of the most interesting historical ethical...
-
What is the solution of the recursive equation T (n) = 0.015625T +n?? (1/4) a) O(n') b) O(n' lg n) c) O(n* Ign) d) O(7*)
-
Which of these compounds are expected to have an absorption maximum in the region of 200 to 400nm in their UV spectra? ) C-CH,CH, b) CH,CH,CH3 c) d) f) CH,CH,OCH,CH3 e) h) g)
-
Explain how mass spectrometry could be used to distinguish between thesecompounds: a) and b) and c) and
-
Suppose your business deals in expensive jewellery. Which inventory system and method should you use to achieve good internal control over the inventory? If your business is a hardware store that...
-
Research the United States Office of Personnel Management (OPM) incident announced in July 2015. Summarize the key aspects of the case and what key/critical pieces of data were found. Then using that...
-
Please view the video below and answer the following questions: 1. What are the 5 stock phases that the speaker suggests you can use to listen better? 2. Provide 3 variations of each of the 5 stock...
-
List two similarities and differences between conventional Western medicine and complementary and alternative health care practices.
-
Unless you drive an electric vehicle, on a day-to-day basis there's a container filled with a highly volatile liquid stashed away somewhere in your car. While the kind of car explosions displayed by...
-
What information should you seek before choosing any health care service or therapy?
-
What systematic changes, if any, should Amy make to improve the job attitudes of the employees of Ohio Strong?
-
As of January 1, 2018, Room Designs, Inc. had a balance of $9,900 in Cash, $3,500 in Common Stock, and $6,400 in Retained Earnings. These were the only accounts with balances in the ledger on January...
-
How might low inflation expectations on the part of the public help to hold down actual inflation?
-
Write a three-dimensional formula for each of the following molecules using the wedge-dashed wedge-line formalism. If the molecule has a net dipole moment, indicate its direction with an arrow, . If...
-
Consider each of the following molecules in turn: (a) Dimethyl ether, (CH3)2O; (b) Trimethylamine, (CH3)3N; (c) Trimethylboron, (CH3)3B; and (d) Dimethylberyllium, (CH3)2Be. Describe the...
-
Analyze the statement: For a molecule to be polar, the presence of polar bonds is necessary, but it is not a sufficient requirement.
-
Problem 1. Non-dimensionalization and phase-space portraits We consider the EOM mx(t) = ax(t) bx (t) (13.1) where x(t) is the position of a particle of mass m. a) What are the dimensions of a and b?...
-
The froghopper Philaenus spumarius is supposedly the best jumper in the animal kingdom. To start a jump, this insect can accelerate at 4.00 km/s over a distance of 2.0 mm as it straightens its...
-
Given a string length of 1.00 0.01 m and a period of 2.00 0.10, what is the uncertainty in gravity? No need to include units, but give them as m/s^2.

Study smarter with the SolutionInn App


