Explain how mass spectrometry could be used to distinguish between thesecompounds: a) and b) and c) and
Question:
Explain how mass spectrometry could be used to distinguish between thesecompounds:
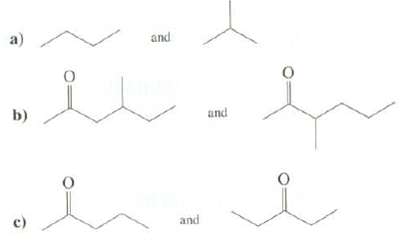
Transcribed Image Text:
a) and b) and c) and
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
a The major fragment In the mass spectrum of butane is an ethyl cation at mz ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain how IR spectroscopy could be used to distinguish between thesecompounds: CH,COCH, and COCH,CH, a) CH,CH,CH,CCH, and CH,CH,CH,CH,CH b) c) CH;CH-CHOCH, and CH,CH-CHCH,COH
-
Explain how IR spectroscopy could be used to distinguish between thesecompounds: and b) and NH2 d d) and and e) ) and
-
Explain how UV spectroscopy could be used to distinguish between thesecompounds. and a) b) and c) and HO and d)
-
On January 15, Tundra Co. sold merchandise to customers for cash of $42,000 (cost $28,500). Merchandise costing $10,500 was sold to customers for $15,800 on January 17; terms 2/10, n/30. Sales...
-
Your boss at your accounting firm wants to start offering accounting services to health care organizations and would like you to brief your entire department on the fundamentals. Your outline would...
-
You are president of the Friends of the Library, a not-for-profit group that raises funds and provides volunteers to support your local library. Every February, you send a report of the previous...
-
In Exercises 1 to 4, it may be helpful to draw a figure such as Figure 5.5. Figure 5.5. Using the normal curve table, determine the area of the standard normal distribution that is greater than the...
-
Scott Snapp contracted with Castle-brook Builders, Inc., which was owned by Stephen Kappeler, to remodel a house. Kappeler estimated that the remodeling would cost around $500,000. Eventually,...
-
Rubber and Steel Company is planning to manufacture a new product. The variable manufacturing costs will be $57 per unit and the fixed costs are estimated to be $6819 The selling price of the product...
-
One of the members of your team continually arrives late for meetings and does not turn drafts of assignments in on time. Choose one of the available theories and indicate how the theory explains the...
-
Predict the major fragments and their m/z that would appear in the mass spectra of these compounds: a) b) c) - d) ) h) g) i)
-
Explain why neopentane shows no molecular ion is its mass spectrum, Predict the structure and m/z for these base ion in its massspectrum. CH3 CH .-, -C- CH3 Neopentane
-
You work for a reputable chemical supply house. An angry customer, Fly Ofterhandle, has called, alleging that a sample of 2,5-hexanediol he purchased cannot be the correct compound. As evidence, he...
-
A certain crying baby emits sound with an intensity of 8.0 x 10-8 W/m2. What is the intensity level due to a set of five such crying babies, all crying with the same intensity? You can neglect any...
-
A race car rounds a curve at 6 0 m / s . The radius of the curve is 4 0 0 m , and the car's mass is 6 0 0 kg . ( a ) What is the centripetal acceleration of the car? ( b ) What is the centripetal...
-
26. An RL series circuit has: 4.8-mH coil, 2.8-2 resistor, 1.0-V battery, ammeter and switch. After the switch is closed for a long time, find the final value of the current. a. 7.4E+1 A b. 3.6E-1 A...
-
Calculate the depth of the ocean if the sound waves from the vessel takes 9 seconds to return to the vessel? a) 13,500 m/s b) 27,000 m/s c) 6,750 m/s d) 4,250 m/s 4
-
A 1 6 0 0 Kg race car exerts 1 4 4 0 0 N of force and accelerates from 0 to 1 0 0 Km / h in 3 . 8 s . What is the force of friction acting on the car?
-
Why is it important for the risk management report form to include who is directly responsible for the risk?
-
The financial statements of Eastern Platinum Limited (Eastplats) are presented in Appendix A at the end of this textbook. Instructions (a) Does East plats report any investments on its statement of...
-
In the 1990s, Dana Mead, the chief executive officer of Tenneco, Inc., quipped, NAIRU is to economics what a Nehru jacket is to fashion: outdated. The relationship between the rate of inflation and...
-
Which compound in each of the following pairs would have the higher boiling point? Explain your answers. (a) (b) (c) (d) (e) (f) (g) (h) Hexane, CH3(CH2)4CH3, or nonane, CH3(CH2)7CH3 (i) OH or HO OH...
-
Predict the key IR absorption bands whose presence would allow each compound in pairs (a), (c), (d), (e), (g), and (i) from Problem 2.46 to be distinguished from each other. In problems 2.46 (a) (c)...
-
The IR spectrum of propanoic acid (Fig. 2.17) indicates that the absorption for the O-H stretch of the carboxylic acid functional group is due to a hydrogen-bonded form. Draw the structure of two...
-
Discuss the incident response steps and the recovery strategy and phases.
-
Compare and contrast planning and innovation with citation and references.
-
How are the tools used for planning, monitoring, and reporting in Six Sigma?

Study smarter with the SolutionInn App


