Convert the following molecule model of hexane, a component of gasoline, into a line-bond structure (gray =
Question:
Convert the following molecule model of hexane, a component of gasoline, into a line-bond structure (gray = C, ivory =H).
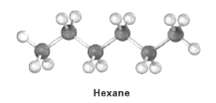
Transcribed Image Text:
св Нехапе
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
01 I...View the full answer

Answered By

Robert Mbae
I have been a professional custom essay writer for the last three years. Over that period of time, I have come to learn the value of focusing on the needs of the clients above everything else. With this knowledge, I have worked hard to become an acclaimed writer that can be trusted by the customers to handle the most important custom essays. I have the necessary educational background to handle projects up to the Ph.D. level. Among the types of projects that I've done, I can handle everything within Dissertations, Project Proposals, Research Papers, Term Papers, Essays, Annotated Bibliographies, and Literature Reviews, among others.
Concerning academic integrity, I assure you that you will receive my full and undivided attention through to the completion of every essay writing task. Additionally, I am able and willing to produce 100% custom writings with a guarantee of 0% plagiarism. With my substantial experience, I am conversant with all citation styles ranging from APA, MLA, Harvard, Chicago-Turabian, and their corresponding formatting. With all this in mind, I take it as my obligation to read and understand your instructions, which reflect on the quality of work that I deliver. In my paper writing services, I give value to every single essay order. Besides, whenever I agree to do your order, it means that I have read and reread your instructions and ensured that I have understood and interpreted them accordingly.
Communication is an essential part of a healthy working relationship. Therefore, I ensure that I provide the client with drafts way long before the deadline so that the customer can review the paper and comment. Upon completion of the paper writing service, the client has the time and right to review it and request any adjustments before releasing the payment.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Convert the following hashed-wedged line formulas into condensed formulas. (a) (b) (c) in NH2 Br H-C Br Br
-
Convert the following condensed formulas into hashed-wedged line structures. (a) (b) CHCI3 (c) (CH3)2NH (d) cHuCHOCL SH ,
-
A gasoline line is connected to a pressure gage through a double-U manometer, as shown in Fig. P1114, if the reading of the pressure gage is 370 kPa determine the gage pressure of the gasoline line....
-
Order check. Write a program that takes three double command-line arguments x, y, and z and prints true if the values are strictly ascending or descending ( x < y < z or x > y > z ), and false...
-
Joanna, age 44, defers $23,000 in a qualified 401(k) plan in 2016. a. What amount must be returned to Joanna and by what date? b. In what year will the amount be taxed? c. What percent of the tax...
-
Give an example of a deadlock taken from politics.
-
The \(f\)-chart is based on \(F_{R}(\tau \alpha)\) and \(F_{R} U_{c}\) values, which can be deduced from a plot of collector efficiency versus \(\left(T_{\text {fin }}-T_{a} ight) / I\). If such a...
-
"In reviewing your previous reports, several questions came to Elizabeth Burkes mind. Use point and interval estimates to help answer these questions. 1. What proportion of customers rate the company...
-
5.a. Is the term representative democracy a contradiction, and, if so, why? (2 marks; word limit: 100 words).
-
The unadjusted trial balance at April 30, 2021, and adjustment data for the month of April 2021 for Garden Designs are presented below: Additional information: 1. $500 of the unearned revenue is to...
-
Draw a line-bond structure for propane, CH3CH2CH3. Predict the value of each bond angle, and indicate the overall shape of the molecule.
-
Draw a line-bond structure for propane, CH3CH = CH2; indicate the hydribization of each carbon; and predict the value of each bond angle.
-
Add the given vectors by components. A = 21.9, A = 236.2 B = 96.7, B = 11.5 C = 62.9, C = 143.4
-
Drug 4 mg I every 3 hours prn pain. Supplied as 10mg/10 ml. Calculate mL per dose?
-
How can one tell many months the positive skew had in it?
-
Show how binary exponentiation can be used to calculate a 128 efficiently. How many multiplications are required?
-
A downton parking lot charges $5 per hour for the first 2 hours, then $2.50 per hour after that. How would I properly equate this?
-
What are m and b in the linear equation 2 + 3x + 5 - 2x = y
-
Eight grocery stores sell the PR energy bar for the following prices: Find the mean, median, and mode for these prices. $1.09 $1.29 $1.29 $1.35 $1.39 $1.49 $1.59 $1.79
-
Use translations to graph f. f(x) = x-/2 +1
-
The molar integral heat of solution s H is defined as the change in enthalpy that results when 1 mole of solute (component 1) is isothermally mixed with N 2 moles of solvent (component 2) and is...
-
Show a Lewis structure for C 2 H 6 O in which both carbons are bonded to the oxygen. What is the geometry of this molecule at the oxygen? Show the direction of the dipole for the molecule,
-
Show a Lewis structure for NO 2 , both oxygens are bonded to the nitrogen.) Show a resonance structure also.
-
Amino acids, from which proteins are formed, exist as ?dipolar ions.? The structure of the dipolar ion of the amino acid alanine is (a) Calculate the formal charges on all of the atoms, except...
-
The following are extracted from the Cash Book of Jungle Trading and its Bank Statement (received from ABC Bank) for the month of April 2020: Cash Book April 2020 RM April 2020 1 Bal b/d 1,500 9...
-
1. Briefly describe FIVE (5) differences between qualitative data and quantitative data. 2. The literature review is an integral part of the research process and make a valuable contribution to...
-
Writing a detailed written assessment that challenges the traditional legal regimes governing the use of military force, addressing key jus ad bellum and jus in bello legal concerns along with their...

Study smarter with the SolutionInn App


