Convert the following representation of ethane, C2H6. Into a conventional drawing that uses solid, wedged, and dashed
Question:
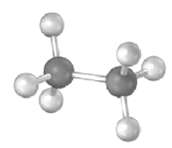
Convert the following representation of ethane, C2H6. Into a conventional drawing that uses solid, wedged, and dashed lines to indicate tetrahedral geometry around each carbon (gray = c, ivory =H).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Indicate the molecular geometry around each carbon atom in the compound H;CCH=CHCH,CCH,COOH
-
Consider the following representation of the reaction 2NO2(g) 2NO(g) + O2(g). Determine the time for the final representation above if the reaction is a. First order b. Second order c. Zero order...
-
The drawing shows a cube. The dashed lines in the drawing are perpendicular to faces 1, 2, and 3 of the cube. Magnetic fields are oriented with respect to these faces as shown, and each of the three...
-
Run experiments to determine the relative costs of Math.exp() and the methods from EXERCISE 1.3.38 for computing e x : the direct method with nested for loops, the improvement with a single for loop,...
-
In 2016, Cao's compensation before his employer's contribution to a SEP is $66,000. Up to what amount can Cao's employer contribute and deduct in 2016?
-
Give structures for compounds A-E. H2CrO (1) CHaMg (2) H20 Cyclohexanol acetone heat (1) 02 12) 2) Mes D (CH1202) (1) Ag20, HO (2) H20* C (C7H2) 2)(C,H120)
-
Air enters a 4-cm-square galvanized steel duct with \(p_{0}=\) \(150 \mathrm{kPa}, T_{0}=400 \mathrm{~K}\), and \(V_{1}=120 \mathrm{~m} / \mathrm{s}\). \(\mu=2.2 \times 10^{-5}\) \(\mathrm{N} \cdot...
-
Presented here are long-term liability items for Evenson Inc. at December 31, 2014. Prepare the long-term liabilities section of the balance sheet for Evenson Inc. Bonds payable (due 2018)..........
-
As with law enforcement and court personnel, corrections, probation, and parole officers are held to a high ethical standard. Corrections, probation, and paroleagencies must be diligent in addressing...
-
Astro Athletic Supply Company specializes in selling sporting goods, gymnasium equipment, and other athletic supplies to high schools and colleges. The company handles both written and telephone...
-
Draw a molecule of chloroform, CHCl3, using solid, wedged, and dashed lines to show its tetrahedral geometry.
-
What are likely formulas for the following substances? (a) GeCl? (b) AlH? (c) CH? Cl2 (d) SiF? (e) CH3NH?
-
Why might these CPI numbers be biased? The Lucky Country reported the following CPI data: June 2010 201.9 June 2011 207.2 June 2012 217.4
-
The revenue cycle is a key element in the management of today's modern hospital. Explain the key elements of the revenue cycle and the issues involved in its management. Also explain why receivables...
-
Find the intermediate points using DDA line algorithm. Assume the starting point (5, 6) and ending point (13, 10). b (6,7), (7,7), (9,9), (9,8), (11,9), (12,9), (12,10), (13, 10). O (6,7), (7,8),...
-
Suppose that you bought a 10-year annual coupon bond with 8% coupon rate and a face value of $1000.At that time the YTM was 6-percent. How much did you pay to buy the bond?
-
When an agent is allowed to make key decisions on behalf of a principal, moral decisions and incentive schemes become critical. Question: Some tools for management to motivate employees to perform at...
-
Ed has a major medical contract that contains a $100 calendar-year deductible. Prior to October of last year, Ed incurred $75 of covered medical expenses. In the last three months of the year, $90 of...
-
What type of algorithm should be used to purchase the XYZ shares given Hardings priority in building the XYZ position and his belief about potential price movements? A. Scheduled algorithm B. Arrival...
-
a. Determine the domain and range of the following functions.b. Graph each function using a graphing utility. Be sure to experiment with the window and orientation to give the best perspective of the...
-
A 20 wt % solution of sulfuric acid in water is to be enriched to a 60 wt % sulfuric acid solution by adding pure sulfuric acid. a. How much pure sulfuric acid should be added? b. If the 20 wt %...
-
Predict which of the following constitutional isomers for the compound that is formed from one atom each of hydrogen, oxygen, and chlorine is more stable: H-CI-: or H-O-CI:
-
Experimental evidence indicates that the two oxygen of acetate ion are identical. Use resonance to explain this observation H H-C-C H Acetate ion : O:
-
Show the direction of the dipoles, if any, of these bonds: (a) C N (b) O N (c) O l (d) C Cl (e) B O (f) C Mg (g) C C (h) C H
-
Describe the impact of Non-Uniform Memory Access (NUMA) on an operating system's memory management strategies, and how an OS can optimize performance in a NUMA-based hardware environment .
-
Explain the concept of system calls in operating systems, and discuss the security implications of exposing system-level operations to user space.
-
Rafner Manufacturing has the following budgeted data for its two production departments. Budgeted Data Assembly Overhead cost $ 1,560,000 $ 1,035,000 Direct labor hours Machine hours 13,000 direct...

Study smarter with the SolutionInn App


