Cyclooctatetraene was first synthesized in 1911 by a route that involved the following transformation: How might you
Question:
Cyclooctatetraene was first synthesized in 1911 by a route that involved the following transformation: How might you use the Hofmann elimination to accomplish this reaction? How would you finish the synthesis by converting cyclooctatriene intoCyclooctatetraene?
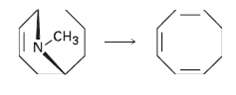
Transcribed Image Text:
CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
N 1 CHI CH3 2 AgO HO 3 heat NBS Br 1 CHI 2 AgO HO 3 ...View the full answer

Answered By

MARK MEKA
Very competent tutor with more than 5 yrs experience ,ready to deliver quality work within stipulated timelines.
Hold Msc procurement and contract management ,am also atrained teacher with Bed (Mathematics and Business studies.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The herbicide acifluorfen can be prepared by a route that begins with reaction between a phenol and an aryl fluoride. Propose a mechanism. NO2 CO2CH3 NO2 co,CH3 NO2 .CO2H DMSO HO F3C F3C CI FC...
-
The amino acid serine is biosynthesized by a route that involves reaction of 3-phosphohydroxypyruvate with glutamate. Propose amechanism. Gutamato a toglutarnto HaN- CH2OPO,2- 3-Phosphoserine C=0...
-
How might you use a local area network in your home? What devices could connect to such a network?
-
Consider a dynamic array where the capacity doubles when additional space is needed and shrinks by 50% when the size falls below 1/4 capacity. What will be the amortized big-O of an append()...
-
What are the three stages of every interview, and which is the most important?
-
The formation of tetrafluoroethylene from its elements is highly exothermic: (a) If a mixture of F2, graphite, and C2F4 is at equilibrium in a closed container, will the reaction go to the right or...
-
Cite several significant events in the development of contemporary internal auditing in the U.S.
-
Henry Horticultural, Ltd., is a leading producer of greenhouse irrigation systems. Currently, the company manufactures the timer unit used in each of its systems. Based on an annual production of...
-
For the year ended December 31, 2019, Dent Co. estimated its allowance for uncollectible accounts using the year-end aging of accounts receivable. The following data are available: Allowance for...
-
The 'estimating trumpet' illustrates tolerances and expectations around levels of estimating accuracy across the life cycle. How do the tolerances resonate with your organisation's expectations...
-
Reaction of anthranilic acid (o-amino benzoic acid) with HNO2 and H2SO4 yields a diazonium salt that can be treated with base to yield a neutral diazonium carboxylate. (a) What is the structure of...
-
When an ?-hydroxy amide is treated with Br2 in aqueous NaOH under Hofmann rearrangement conditions, loss of CO2 occurs and a chain-shortened aldehyde is formed. Propose a mechanism. NH2 NH3 Br2...
-
At the bottom of all its February sales receipts, Seifert Stores Inc. printed $2-off coupons. The coupons may be redeemed March 1April 30. Seifert Stores Inc. accepts only cash, Mastercard, or Visa....
-
Factor completely; simplify if possible. 9r2+24r +16=
-
Factor completely. 10v7 +32v6 +6v5
-
Multiply. 45x-25 2x+14 6x+42 9x-5 Simplify your answer as much as possible.
-
Simplify. 3 X 205x X 2 16-x
-
Solve the proportion and check. 96 10 5 *19
-
Sage Inc.s staff of accountants finished preparing the financial statements for the most recent year and will meet next week with the companys CEO as well as the Director of Investor Relations and...
-
Write the binomial probability in words. Then, use a continuity correction to convert the binomial probability to a normal distribution probability. P(x 110)
-
What is the percent ionization of an acid? Explain what happens to the percent ionization of a weak acid as a function of the concentration of the weak acid solution.
-
Write a complete reaction mechanism for the addition of HCl to 1-butyne (Problem 3.59a).
-
For the structures of nerolidol and 4,8-dimethyl-1,3,7-nonatriene in Problem 3.40, provide the products for each compound after: a. Hydrogenation (3 moles H2, Pt catalyst) b. Addition of 1 mole of...
-
When 1-butyne is treated with followed by H2O2 and OH, the product isolated is the aldehyde butanal : CH3 CH,CH,CH,--
-
Calculating Present Values Imprudential, Inc., has an unfunded pension liability of $645 million that must be paid in 25 years. To assess the value of the firm's stock, financial analysts want to...
-
Describe the operating activities of each company noting similarities and difference between COCA COLA & PEPSICO. Identify two economy wide factors and industry wide factors that could impact on the...
-
The trial balance for a company listed the following account balances at December 31, Year 1, the end of its fiscal year: cash, $36,000; accounts receivable. $31,000; Inventory, $45,000; equipment...

Study smarter with the SolutionInn App


