When an ?-hydroxy amide is treated with Br2 in aqueous NaOH under Hofmann rearrangement conditions, loss of
Question:
When an ?-hydroxy amide is treated with Br2 in aqueous NaOH under Hofmann rearrangement conditions, loss of CO2 occurs and a chain-shortened aldehyde is formed. Propose a mechanism.
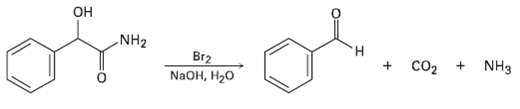
Transcribed Image Text:
Он NH2 н NH3 Br2 NaOH, H20 CO2 +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
NH H 32533 H NH Br NaOH HO NH3 NH3 Hofm...View the full answer

Answered By

Vineet Kumar Yadav
I am a biotech engineer and cleared jee exam 2 times and also i am a math tutor. topper comunity , chegg India, vedantu doubt expert( solving doubt for iit jee student on the online doubt solving app in live chat with student)
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
When the following compound is treated with Br 2 in the presence of a Lewis acid, one product predominates. Determine the structure of that product. Br FeBr3 ?
-
When 1,3-diphenyl-2-propanone is treated with Br2 in acid, l,3-dibromo-l,3-diphenyl-2-propanone is obtained in good yield. On further characterization, however, this product proves to have a very...
-
Compound X is treated with Br 2 to yield meso-2,3- dibromobutane. What is the structure of compound X?
-
In Problem 12.4 on page 423, you used the percentage of alcohol to predict wine quality. The data are stored in VinhoVerde. From the results of that problem, b 1 = 0.5624 and Sb 1 = 0.1127. a. At the...
-
How does a structured interview differ from an open ended interview?
-
How does conventional retained earnings differ from entity equity under the Anthony conception of the entity theory?
-
Are Carbon or Steel Bikes Faster? Dr. Jeremy Groves was interested in whether his carbon bike or his steel bike led to a shorter commute time. To answer this, he flipped a coin each day to randomly...
-
Marienau Suppliers had the following transactions: Mar. 1 Sold merchandise on account to G. Perez $5,000. 20 G. Perez gave a $5,000, 90-day, 6% note to extend time for payment. 30 G. Perez's note is...
-
Identify any five stakeholders who would be interested in Grassland s financial information and explain why and how each would use the information.
-
Which bond in Exhibit 1 most likely has the lowest effective convexity? A. AI bond B. BI bond C. CE bond Jules Bianchi is a bond analyst for Maneval Investments, Inc. Bianchi gathers data on three...
-
Cyclooctatetraene was first synthesized in 1911 by a route that involved the following transformation: How might you use the Hofmann elimination to accomplish this reaction? How would you finish the...
-
The following transformation involves a conjugate nucleophilic addition reaction (Section 19.13) followed by an intra molecular nucleophilic acyl substitution reaction (Section 21.2). Show...
-
Sometimes, the desire to maintain group harmony overrides the importance of making sound decisions. When that occurs, team members are said to engage in groupthink. According to Michael Useem, a...
-
How did historical events affect American literature?(Sample question:How did the Civil War affect American Literature?) How did the Civil War transform American literature? Does literature affect...
-
Write in 1 - 2 Paragraph the effect of European Literature in world literature. Explain its relevance/Significance to you a 21st century literature student.
-
Write a complex program in java spring booth to show the weather forecasting.
-
What is the importance of studying world literature? What do you is the prime contribution of any remarkable piece of literature to your course? How do you think each piece of literature reveal the...
-
1. What is the difference between the literature of the past and the literature of the present? 2. in your opinion, Should literature of the past be rewritten in present-day language for today's...
-
Johnson and Bates run a small business and are worried about their accounts receivable spiralling out of control. a Advise them of the steps they can take to manage credit and recover outstanding...
-
What services are provided by the provincial and territorial governments?
-
Find the [C 6 H 6 O 6 2- ] of the 0.100 M ascorbic acid (H 2 C 6 H 6 O 6 ) solution in Example 17.17. Example 17.17 Find the pH of a 0.100 M ascorbic acid (H 2 C 6 H 6 O 6 ) solution.
-
As discussed in "A Word About . . . Petroleum, Gasoline, and Octane Number" on p. 103, isomerization is often used to convert one alkane to a more substituted alkane. Suggest the highly branched...
-
Why, in general, is a sigma bond between two atoms stronger than a pi bond between the same two atoms?
-
Write an equation for the reaction of bromine at room temperature with a. Propene b. 4-methylcyclohexene
-
An analysis of the activities needed to produce each product has been conducted. In addition, estimates have been developed. These are in the table below. Match the OH rate with the activity for each...
-
Service business 1. You are required to form a small business (Service business) 2. Explain the name and nature of the business, location, mission and objectives of the company. Assume the business...
-
Cost of utilities Cost per month (In dollars) $20,000 4. The cost of utilities depends on how many wards the hospital needs to use during a particular month. During months with activity under 2,000...

Study smarter with the SolutionInn App


