Diethylstilbestrol (DES) has estrogenic activity even though it is structurally unrelated to steroids. Once used as an
Question:
Diethylstilbestrol (DES) has estrogenic activity even though it is structurally unrelated to steroids. Once used as an additive in animal feed, DES has been implicated as a causative agent in several types of cancer. Show how DES can be drawn so that it is sterically similar toestradiol.
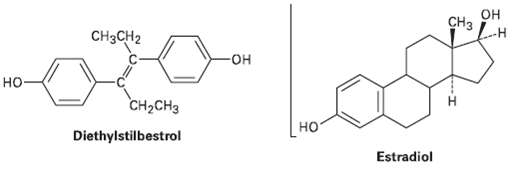
Transcribed Image Text:
Cнз он CH3CH2 но CH-CHз но Diethylstilbestrol Estradiol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (10 reviews)
HO H Estradiol HT CH3OH H CH3 Logo HC D...View the full answer

Answered By

Mario Alvarez
I teach Statistics and Probability for students of my university ( Univerisity Centroamerican Jose Simeon Canas) in my free time and when students ask for me, I prepare and teach students that are in courses of Statistics and Probability. Also I teach students of the University Francisco Gavidia and Universidad of El Salvador that need help in some topics about Statistics, Probability, Math, Calculus. I love teaching Statistics and Probability! Why me?
** I have experience in Statistics and Probability topics for middle school, high school and university.
** I always want to share my knowledge with my students and have a great relationship with them.
** I have experience working with students online.
** I am very patient with my students and highly committed with them
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Is VCW Inc. a social enterprise even though it is a for-profit business? On what do you base your answer? Is Cheryl Womack a social enterpriser?
-
Show how dimedone can be synthesized from malonic ester and 4-methyl-3-penten-2-one (mesityl oxide) under basic conditions. OH Dimedone
-
Show how 1-butanol can be converted into the following compounds: a. CH3CH2CH2CH2Br b. c. CH3CH2CH2CH2OCH3 d. CH3CH2CH2CH2NHCH2CH3 e. CH3CH2CH2CH2SH f. CH3CH2CH2CH2C==N CH CH2CHCH2OCCH2CH,
-
Simplity each of the follewing ratios. f r 15 15kg:350 g 0.45:085 ( 580 ml: L121:104 m/ 40 033:063: 18
-
1. Why would Alibaba choose to list in the U.S. and not in the Hong Kong Exchange? 2. The Hong Kong Exchange rejected Alibaba's application to list on it because of its corporate governance...
-
Determine the saturation current (ICsat) f°r tne network of Fig. 4.112. In Figure 4.112 20 V 2.4 k 510 k Va cr- 100 1.5 k
-
The following are the data on time taken by a computer engineer to assemble 8 computers each for 3 types of mother boards. Use the \(H\) test at the 0.05 level of significance to test whether there...
-
Rita Lane is the accountant for Outdoor Living, a manufacturer of outdoor furniture that is sold through specialty stores and Internet companies. Lane is responsible for reviewing the standard costs....
-
BMX Company has one employee. FICA Social Security taxes are 6.2% of the first $137,700 paid to its employee, and FICA Medicare taxes are 1.45% of gross pay. For BMX, its FUTA taxes are 0.6% and SUTA...
-
a. To simplify the analysis, assume for now that there is virtually no turnaround time between flights so the next flight can begin as soon as the current flight ends. (If an immediate next flight is...
-
Diterpenoids are derived biosynthetically from geranyl-geranyl diphosphate (GGPP), which is usd1 biosynthesized by reaction of farnesyl diphosphate with isopentenyl diphosphate. Show the structure of...
-
Propose a synthesis of diethylstilbestrol (Problem 27.44) from phenol and any other organic compound required.
-
Why do organizations often combine organization by function and organization by division? What advantages do organizations get from this more complex hybrid form? Organizational Design by Division...
-
The current spot rate between the euro and dollar is 1.0939/$. The annual inflation rate in the US is expected to be 2.83 percent and the annual inflation rate in euroland is expected to be 2.31...
-
Develop a C# console application that simulates a vending machine. Write a method in C# that converts a number from binary to decimal format. Create a simple To-Do List desktop application using...
-
What is the free Cu 2 + concentration if 0 . 0 2 0 M Cu 2 + solution is mixed with an equal volume of 4 . 0 M NH 3 ? Kf for [ Cu ( NH 3 ) 4 ] 2 + is 1 . 1 \ times 1 0 1 3 .
-
Payback, NPV, and MIRR Your division is considering two investment projects, each of which requires an up-front expenditure of $27 million. You estimate that the cost of capital is 12% and that the...
-
solve all the attachments What is implied by a serializable request for at least two exchanges? [2 marks] (b) Explain how timestamp requesting (TSO) upholds segregation. [5 marks] (c) Draw and make...
-
Complete the approximation argument for Lvy's arc-sine law from \(\S 8.9\) : a) Show, by a direct calculation, that \(v_{n, \lambda}(x)\) converges as \(n ightarrow \infty\). Conclude from (8.16)...
-
A line l passes through the points with coordinates (0, 5) and (6, 7). a. Find the gradient of the line. b. Find an equation of the line in the form ax + by + c = 0.
-
The evaporation of a 120-nm film of n-pentane from a single crystal of aluminum oxide is zero order with a rate constant of 1.92 * 10 13 molecules/cm 2 s at 120 K. a. If the initial surface coverage...
-
Starting with -d-glucose and using acid (H1) as a catalyst, write out all of the steps in the mechanism for the mutarotation process.
-
Lactose exists in a and b forms, with specific rotations of 192.6 and 134, respectively. a. Draw their structures.
-
Oxidation of either d-erythrose or d-threose with nitric acid gives tartaric acid. In one case, the tartaric acid is optically active; in the other, it is optically inactive. How can these facts be...
-
www The sugar cane industry is a big industry on Hawaii. Raw sugar cane is first cut from the fields, then chopped and shredded. The raw cane contains 15 wt% sucrose, 25 wt% solids, and water, along...
-
What does a manager who subscribes to Theory X believes ? Explain thoroughly.
-
Keri, in forming a new corporation, transfers land to the corporation in exchange for 100 percent of the stock of the corporation. Keri's basis in the land is $295,000, and the corporation assumes a...

Study smarter with the SolutionInn App


