Draw as many resonance structures as you can for the following species. Adding appropriate formal charger to
Question:
Draw as many resonance structures as you can for the following species. Adding appropriate formal charger to each:
(a) Nitromethane,
(b) Ozone,
(c) Diazomethane,
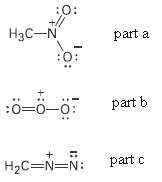
Transcribed Image Text:
:0: +// H3C-N part a :0: part b :0=0-0: part c H2C=N=N: :o:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a c 0 HC...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw as many resonance structures as you can for the followingspecies: (b) :NH2 H2N-C=NH2 (c) (a) :0: H --- () HC $H2 (e) H %H
-
Suggest the best syntheses that you can for the following amines, beginning each with any organic compounds that do not contain nitrogen. (a) Butanamine; (b) N-methylbutanamine; (c)...
-
Draw contributing resonance structures for each of the following species, and rank the structures in order of decreasing contribution to the hybrid: a. b. c. d. e. f. CH3C-CH CHCH3 CH3 0 CH3COCH3 +OH...
-
In a plant producing multiple products, would absorption costing overcost or undercost the more complex products? Why?
-
In 2016, Bianca earned a salary of $164,000 from her employer. Determine the amount of FICA taxes and Medicare taxes withheld from her salary.
-
A Ca 2+ ion-selective electrode was calibrated in metal ion buffers with ionic strength fixed at 0.50 M. Using the following electrode readings, write an equation for the response of the electrode to...
-
In a transactional data, what is the effect of aggregating similar items into a broader product category? a. Item granularity does not matter as one can simply adjust the minimum support threshold....
-
The Lifestyle Clothes Company produced 24,000 units during April of the current year. The Cutting Department used 4,000 direct labor hours at an actual rate of $11.20 per hour. The Sewing Department...
-
What are the challenges in integrating process simulation with real-time process control and automation? How can digital twins and cyber-physical systems support the seamless integration of...
-
QUESTION 1 ?Is this statement true or false??Even if plagiarism is discovered years from now, a university can withdraw a qualification that was awarded to a student? A. True B. False 1 points ?...
-
Which of the following pairs represent resonance structures? (a) CH3C=N-O: and CH3C=N-o: (b) :0: :C C0: and (d) (c) : and :CH2-N CH2=N NH3 and NH2
-
Carbocations, ions that contain a trivalent, positively charged carbon atom, react with water to give alcohols. How can you account for the fact that the following carbocation gives a mixture of two...
-
Use thermodynamic data from Appendix D to calculate a theoretical voltage of the silverzinc button cell described on page 893. TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z...
-
1.Your client has an asset of USD 1 million with a liability of USD 500,000. 2.The maturity of the liability is 20 years with the yield to maturity of 4%. 3.The client's investment horizon is 3...
-
Recently, you needed money and agreed to sell a car you had inherited at a price of $45,000 , to be paid in monthly payments of $1,300 for 45 months . What (annual) interest rate did you charge for...
-
Adam works as a financial analyst with General Motors. He was asked by his boss to evaluate two potential projects. The initial cost of project 1 is $300,000 and is expected to produce cash flows of...
-
A gear with a radius of 9.0 inches makes 15 revolutions every four seconds. Find the linear and angular velocities of a point on the outer edge of the gear. Give your answers in both exact and...
-
The Geneva mechanism is used in a packaging system to convert constant angular motion into intermittent angular motion. The star wheel A makes one sixth of a revolution for each full revolution of...
-
Determine the most appropriate currency management strategy for Bhatt. Justify your response. Following analysis of Indian economic fundamentals, C&Ms currency team expects continued stability in...
-
Prove the formula for (d/dx)(cos-1x) by the same method as for (d/dx)(sin-1x).
-
Using the Redlich-Kwong equation of state, compute and plot (on separate graphs) the pressure of nitrogen as a function of specific volume at the two temperatures: a. 110 K b. 150 K
-
Explain in which solvent these reaction are faster: a) HC Br in CHOH or CH-CHOH I b) CHCHCHCH + OH in CHOH or 50% CHOH 50% HO CHICHICH c) CHCHCH + C=N: in CHCHOH or DMSO
-
Explain whether these reactions follow an SN1 or an SN2 mechanism. CH3 T a) CH3-C-Br + CHCO CH3 b) OMS d) + SH CH3 c) CH CH-C-0 T CH3 DMF CHCH-Br + CHOH + CH CO + CHCHOH CH,COH 1-BUOH CHOH CHCCH EtOH
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Solve these equations Sy = b with 81, 82, 83 in the columns of S: 0 0 10 Y2 100 0 1998-8-1988-8 and S is a sum matrix. The sum of the first 5 odd numbers is ][
-
Active Life Ltd has decided to manufacture a new line of running shoes based on a $50,000, two-year feasibility study. The shoes will sell for $85 a pair and has a variable cost of $30 a pair. It is...
-
Arch Airway expects to purchase 5.6 million gallons of jet fuel in three months and decides to use heating oil futures to hedge the fuel price risk. A regression has been performed: The dependent...

Study smarter with the SolutionInn App


