Which of the following pairs represent resonance structures? (a) CH3C=N-O: and CH3C=N-o: (b) :0: :C C0:
Question:
Which of the following pairs represent resonance structures?
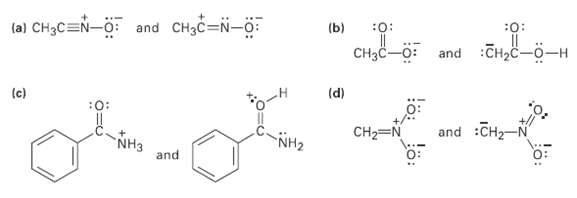
Transcribed Image Text:
(a) CH3C=N-O: and CH3C=N-o: (b) :0: :Cнас —о—н Cнас—0: and (d) (c) : and :CH2-N CH2=N NH3 and NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
Pairs a and d resonance structu...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following pairs of structures represent resonanceforms? (b) :0: (a) :0: and and (d) :: (c) :0: :0: and and
-
Which of the following structures represent the same compound? Which ones represent different compounds? (a) (b) (c) (d) (e) (f) (g) Name the structures given in Problem 3-33, parts (a), (c), (e),...
-
Which of the following pairs of structures represent the same enantiomer, and which represent different enantiomers? (b) Br Br C (a) CN - N - - r H3CC-CN Br CN (c) (d) CH C g H- H2N CH - -H -...
-
What problems are associated with tracking historical costs?
-
Determine the additional Medicare taxes for these individuals. a. Mario, who is single, earns wages of $440,000 in 2016. b. George and Shirley are married and file a joint return in 2016. During the...
-
From the following potentials, calculate the activity of Cl - in 1 M KCl. E(calomel electrode) = 0.268 V E(calomel electrode, 1 M KCl) = 0.280 V
-
Which of the following statements about lift is false? a. Lift can be interpreted as how much more likely the antecedent and consequent itemsets occur together than if they were totally unrelated. b....
-
Data for Whitlock Company are presented in P13-3A. In P13-3A, The income statement of Whitlock Company is presented here. Additional information: 1. Accounts receivable increased $200,000 during the...
-
Now that we're talking about Operant Conditioning, learner's choice, where they emit a behavior (response) first, in order to get/avoid a stimulus (reinforcement/punishment), let's take it slow and...
-
What risks might financial institutions face by funding long-run loans such as mortgages to borrowers (often at fixed interest rates) with short-term deposits from savers?
-
Identify the acids and bases in the followingreactions: CH2 (a) CH3 + Ht TICIA () + TICI, CH "CH (c) + NaH +. Na+ 2 (d) +
-
Draw as many resonance structures as you can for the following species. Adding appropriate formal charger to each: (a) Nitromethane, (b) Ozone, (c) Diazomethane, :0: +// H3C-N part a :0: part b...
-
What is the difference between a hard link and a symbolic link? Give an advantage of each one.
-
Currently, 5-year annual corporate bonds with an AA rating have an average credit spread of 150 basis points. The yield to maturity of 5-year Treasury annual coupon bonds is currently 0.8%. Estimate...
-
Calculate accurate financial ratios to assess the business's current financial health. Specifically, calculate the following ratios: Working capital Current ratio Debt ratio Return on equity based on...
-
What are the experiences that occur in the Hospitality & Tourism Industry that risk personal security and loss of physical property? How was the situation handled by the organization? How should the...
-
Your work colleague has estimated a regression to predict the monthly return of a mutual fund (Y) based on the return of the S&P 500 (X). Your colleague expected that the "true" relationship is Y =...
-
Major Baker is assessing a Service Member for concussion. Major Baker is at 15B, Months in Reverse Order. The Service Member has just repeated the months in reverse order but missed September. What...
-
Calculate the contribution of foreign currency to the Bhatt accounts total return. Show your calculations. Darden meets with Bhatt and learns that Bhatt will be moving back to his home country of...
-
A parking lot charges $3 for the first hour (or part of an hour) and $2 for each succeeding hour (or part), up to a daily maximum of $10. (a) Sketch a graph of the cost of parking at this lot as a...
-
Repeat Problem 5.10 assuming that nitrogen is described by the Peng-Robinson equation of state. Problem 5.10 The Quick Fill bicycle tire filling system consists of a small (2 cm diameter, 6.5 cm...
-
Show the products and the mechanisms of the following reactions. Don't forget to use curved arrows to show the movement of electrons in each step of the mechanism. CI a) CHCHCHCH SH SN2 OT's c)...
-
Show all of the steps in the mechanism for this reaction: CH 3 CH, C-CI+ CHOH CH3 CH CH3-C-OCH3 + HCI T CH3
-
Explain why the carbocation shown in Figure 8.8 has a longer lifetime than it does under the conditions shown in Figure 9.8.
-
Find the determinant of the linear transformation T(M): [3] M from the space V of 22 upper triangular matrices to V.
-
1. A car accelerates from rest to 16.21 meters per second in 7.66 seconds. What is the car's rate of acceleration in meters per second squared? Round your answer to two decimal places. 2. If the...
-
The debits to Work in Process-Roasting Department for Morning Brew Coffee Company for August, together with information concerning production, are as follows: Work in process, August 1, 800 pounds,...

Study smarter with the SolutionInn App


