Draw curved-arrow mechanisms for the reactions given in Fig. P18.73.
Question:
Draw curved-arrow mechanisms for the reactions given in Fig. P18.73.
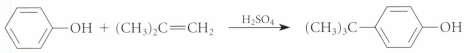
Transcribed Image Text:
он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Protonation of 2methylpropene gives t...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Write out the steps in the reaction mechanisms for the reactions given in eq. 8.20. CHO_ HOCH2CH2OCH, 2-methoxyethanol H+ CH-CH HOCHCH OH HOCH CH2OCH CH OH diethylene glycol
-
Although the synthesis of heterocyclic rings was not discussed in the text, many such syntheses employ reactions that are similar or identical to reactions in other parts of the text. Give...
-
Using the curved-arrow notation, write in detail the mechanisms for the reactions in (a)Eq. 26.41a
-
Markku Antero Oy produces perfume. To make this perfume, Markku Antero uses three different types of fluid. Tartarus, Erebus and Uranus are used in standard proportions of -14-0, -3- 10 and -3- 10,...
-
Listed below are accounts to use for transactions (a) through (j), each identified by a number. Following this list are the transactions. You are to indicate for each transaction the accounts that...
-
A vertical cylinder with a heavy piston contains air at a temperature of 300 K. The initial pressure is 200 kPa, and the initial volume is 0.350 m3. Take the molar mass of air as 28.9 g/mol and...
-
A & R Industrial Supply shows the following financial statement data for 2008, 2009, and 2010. Prior to issuing the 2010 statements, auditors found that the ending inventory for 2008 was understated...
-
Coastal Computer operates two retail outlets in Oakview, one on Main Street and the other in Lakeland Mall. The stores share the use of a central accounting department. The cost of the accounting...
-
How do social identity processes, such as categorization, identification, and comparison, influence team cohesion and performance within complex organizational environments ?
-
An analyst must decide between two different forecasting techniques for weekly sales of roller blades: a linear trend equation and the naive approach. The linear trend equation is Ft = 124 + 2 t, and...
-
When 1, 3, 5-trinitrobenzene [NMR: 9.1 (s)] is treated with Na+ CH3O-, an ionic compound is formed that has the following NMR spectrum: 3.3 (3H, s); 6.3 (1H, t, J = 1 Hz); 8.7 (2H, d, J = 1Hz)....
-
In the conversion shown in Fig. P18.77, the Diels-Alder reaction is used to trap a very interesting intermediate by its reaction with anthracene. From the structure of the product, deduce the...
-
Tiger Inc. has the following year-end account balances: Sales Revenue $928,900; Interest Income $17,500; Cost of Goods Sold $406,200; Operating Expenses $129,000; Income Tax Expense $55,100; and...
-
On January 1, 2025, Cheyenne Inc. issued $5018000, 9% bonds for $4798000. The market rate of interest for these bonds is 10%. Interest is payable annually on December 31. Cheyenne uses the...
-
Otto Enterprises has a bond issue outstanding with an annual coupon rate of 6 percent that matures in 9 years. The bond is currently priced at $1,046.92 and has a par value of $1,000. Interest is...
-
After your victory in the Financial Markets inter-university tournament last month, you are ready to enter the financial scene. You join a group of other SUSS students to compete against the best...
-
An IPv4 packet has arrived its destination with a following header: 4 8 1.) 11.) 111.) 10 75000 89 0 2500 X 925 172.168.10.1 192.168.1.1 How many bytes of data are being carried by this packet? How...
-
Sierra Company Adjusted Trial Balance December 31 Account Title Cash Prepaid insurance Notes receivable (due in 5 years) Buildings Accumulated depreciation-Buildings Accounts payable Debit Credit $...
-
Carranza, a nonexempt salesperson with Verent Enterprises, earns a base annual salary of $30,000 with a standard 40-hour workweek. In addition, a 3 percent commission on all sales during the pay...
-
What are the typical record-at-a-time operations for accessing a file? Which of these depend on the current file record?
-
Bond strengths can be used to estimate the relative stability of isomers that have different bonds. The isomer that has the larger total bond energy is more stable. One of the following isomers is...
-
One of the isomers of C 5 H 12 reacts with Cl 2 in the presence of light to produce three isomers of C 5 H 11 Cl: This reaction replaces am one of the hydrogen?s of C 5 H 12 with a Cl. What arc the...
-
On reaction with Cl 2 in the presence of light, an unknown compound with the formula C 5 H 10 gives only one isomer of C 5 H 9 Cl (see problem 2.39). What is the DU of the unknown compound? Show the...
-
Using the right-hand rule, in which direction will the magnetic force act on a positively charged particle that is moving to the left and experiencing a magnetic field straight ahead? a. Using the...
-
4 The manager of the Fleet division of Potlatch Automotive is evaluated on her division's return on investment and residual income. The company requires that all divisions generate a minimum return...
-
At the end of January of the current year, the records of Donner Company showed the following for a particular item that sold at $16 per unit: Transactions Units Amount Inventory, January 1 Purchase,...

Study smarter with the SolutionInn App


