Ethers can be cleaved by treatment with strong acids. Show all of the steps in the mechanism
Question:
Ethers can be cleaved by treatment with strong acids. Show all of the steps in the mechanism for this reaction and explain why these products are formed rather than iodomethane and2-methyl-2-butanol:
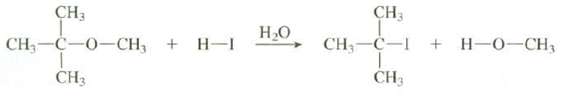
Transcribed Image Text:
CH3 CH3 Н.о CH, —С—0—сH, + н-1 CH,-C-I + H-O-CH, CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (19 reviews)
The conditions for this reaction are favorable fo...View the full answer

Answered By

Keziah Thiga
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagirism), well-researched and critically analyzed papers.
4.90+
1504+ Reviews
2898+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show all of the steps in the mechanism for this reaction and explain the regiochemistry of theaddition: Br Br + HBr
-
Show all of the steps in the mechanism for this reaction, what other products would you expect to beformed? CH3 Br CH3 . CH,O -CH,CH CH.C- C=C CH,CH3 CH3 .
-
Show all of the steps in the mechanism for this reaction: CH 3 CH, C-CI+ CHOH CH3 CH CH3-C-OCH3 + HCI T CH3
-
Tony acquired 1,000 shares in X Co (a resident public company) for $10 each in August 2000. In January this year X Co returned $7 of capital to its shareholder in respect to each share they held. The...
-
The following data relate to Twilight Manufacturing Ltd: During July, 133,500 units were completed and transferred out. Work in process, 1 June: Direct...
-
During the 20072008 NCAA college basketball seasons, mens basketball teams attempted an all-time high number of 3-point shots, averaging 19.07 shots per game (Associated Press Sports, January 24,...
-
Suppose Apex Electrical Supplys Inventory account showed a balance of \($43,000.\) A physical count showed \($41,800\) of goods on hand. To adjust the inventory account, Apex Electrical Supply, Inc.,...
-
A firm wishes to minimize annual inventory costs. The firm uses the EOQ model to determine the cost-minimizing order quantity and the reorder point. Annual demand, units 22,100 Item cost, $ per unit...
-
For each of the following depreciable assets, determine the missing amount. Abbreviations for depreciation methods are SL for straight-line and DDB for double-declining-balance. Note: Do not round...
-
1. Work up a buying plan to use when buying from Ghuman's. 2. How should Hughes distribute the allotted open-to-buy dollars among the available sizes, colors, and fabrics? Synopsis: A department...
-
Explain why this primary halide reacts very rapidly under condition that is favorable for the SN1 mechanism: CH3 O CH2 Cl
-
The reaction of a compound with silver nitrate in ethanol is used as a chemical test to determine if the compound is an alkyl halide. The formation of a precipitate of the silver halide constitutes a...
-
Think about a job that youve held in which you felt very low levels of psychological empowerment. What could the organization have done to increase empowerment levels?
-
Suppose a bank currently has $240,000 in deposits and $23,000 in reserves. The required reserve ratio is 13%. If at the end of the day, there is an unexpected withdrawal of $4,000 in reserves, what...
-
The growth rate for the firm's common stock is 7%. The firm's preferred stock is paying an annual dividend of $5. What is the preferred stock price if the required rate of return is 8%?
-
1) Calculate the magnitude of the electric field generated (8.4x10^0) m from an electron, giving your answer in N/C to 2 significant figures. Magnitudes must be positive. 2) A non-conducting...
-
What role does the span of control play in shaping managerial effectiveness, and how does it relate to organizational hierarchy and employee autonomy ?
-
The last dividend paid by Klein, Inc. was $2.20. Klein's growth rate is expected to be 10 percent for two years, after which dividends are expected to grow at a rate of 6 percent forever. The...
-
What is the largest frequency whose vibrations can be measured by an undamped accelerometer of natural frequency \(200 \mathrm{rad} / \mathrm{s}\) if the error is no more than 1 percent?
-
The financial statements of Eastern Platinum Limited (Eastplats) are presented in Appendix A at the end of this textbook. Instructions (a) Does East plats report any investments on its statement of...
-
How much money is spent on the mortgage? Monthly Budget = $4,000 UTILITIES 10% FOOD 25% CAR 12% SAVINGS MORTGAGE 40%
-
1-Butanol is prepared commercially by a route that begins with an aldol reaction. Show the steps that are likely to be involved.
-
Show how you would synthesize the following compound using an aldolreaction:
-
Which of the following compounds can probably be prepared by a mixed aldol reaction? Show the reactants you would use in eachcase. - (b) (a) (c) n H-CH CH H CH
-
A body temperature of 97.0 F is slightly lower than the "normal" value but is still considered acceptable as an individual's temperature can have variable norms. What would this temperature equate to...
-
Thunder clouds are polarised. For instance if the bottom of the cloud is negatively charged, the top is positively charged. The negatively charged bottom of a thundercloud attracts the positive...
-
You toss a conductive open ring of diameter d = 3.95 cm up in the air. The ring is flipping around a horizontal axis at a rate of 7.25 flips per second. One flip is a full rotation. At your location,...

Study smarter with the SolutionInn App


