Explain which route would provide a better synthesis of theseethers: CH3 CH, CH3 CHI + CH,CO a)
Question:
Explain which route would provide a better synthesis of theseethers:
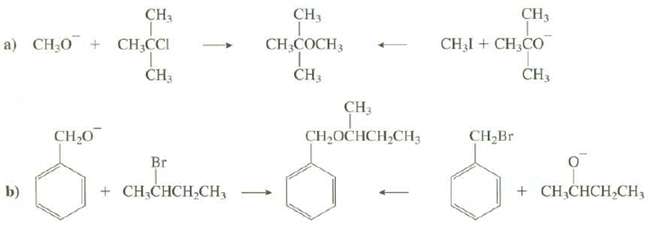
Transcribed Image Text:
CH3 CH, CH3 CHẠI + CH,CO a) CH.O + CH,CCI CH3 CH,COCH3 ČH3 CH3 CH, CH,OCHCH,CH; CH,Br CH,0 Br + CH;CHCH,CH3 b) + CH;CHCH,CH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a The right route is better because methyl iodide cannot undergo an e...View the full answer

Answered By

Bree Normandin
Success in writing necessitates a commitment to grammatical excellence, a profound knack to pursue information, and a staunch adherence to deadlines, and the requirements of the individual publication. My background comprises writing research projects, research meta-analyses, literature reviews, white paper reports, multimedia projects, reports for peer-reviewed journals, among others. I work efficiently, with ease and deliver high-quality outputs within the stipulated deadline. I am proficient in APA, MLA, and Harvard referencing styles. I have good taste in writing and reading. I understand that this is a long standing and coupled with excellent research skills, analysis, well-articulated expressions, teamwork, availability all summed up by patience and passion. I put primacy on client satisfaction to gain loyalty, and trust for future projects. As a detail-oriented researcher with extensive experience surpassing eight years crafting high-quality custom written essays and numerous academic publications, I am confident that I could considerably exceed your expectations for the role of a freelance academic writer.
5.00+
7+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
-
Explain which of these reactions would provide a better synthesis of2-pentene: Br CH,OH CH,CH,CHCH,CH, + CH;0 CH,CH=CHCH,CH3 Br CH, CH CH=CHCH CH, CH,CHCH,CH,CH; + CH,0
-
Provide a retrosynthetic analysis and synthesis for each of the following compounds. Permitted starting materials are phenylmagnesium bromide, oxirane, formaldehyde, and alcohols or esters of four...
-
Evaluate the integral (4e* + 2 In (2))dx.
-
Some businesses use only approximations when estimating cost functions. What are some possible reasons for this?
-
The study in Example 5 found that 32.0% of all men in the United States ages 50 and older have arthritis. You randomly select 75 men in the United States who are at least 50 years old and ask them...
-
Describe the relationship between change control procedures and the configuration control element of CM.
-
1. Characterize the product space for mobile phones when Iridium began. 2. What trends did Nokia pursue as it designed mobile phone products in the late 1990s? 3. What might a more proactive Motorola...
-
Does the private security employee still have the equivalent power of a private citizen? Should they? Why or why not
-
Your brother has just started a new job as the Controller of an IESBA restricted audit client. You do not serve on the audit engagement. What steps must you take to ensure your independence is not...
-
Diphenhydramine can also be synthesized by heating bromo diphenyl methane with the amino alcohol shown here. Offer a reason why the oxygen, rather than the nitrogen, of this compound acts as the...
-
Suggest a synthesis of these ethers starting with an alcohol and an alkylhalide: OCH CH,CH a) CH,OCH,CH,CH,CH, b) c)
-
Refer to Problem 78. The costs of the four brands are Brand A, $1.50; Brand B, $3.00; Brand C, $3.75; and Brand D, $2.25. Which of the solutions to Problem 78 would minimize the cost of the rat food?...
-
Merger Analysis FIN490 | Understanding Merger Transactions Links to an external site. and FIN490 | Valuing Synergies in Transactions Links to an external site.. In your journal, Explain what the...
-
If you extend the financial statements, and performance, what do you expect to be the financial position of Horniman in 2016? Explain. Please show a excel forecasting for 2016 Income statement,...
-
Consider the region D = {(x, y) = R | x 2, (x 2) + y 1}. 1 Using the polar coordinates transformation with center at the point (1, 0) shows that D is the image of a simple region in the plane (r, 0)....
-
The mean inter-arrival times of professors and students to a restaurant inside the campus is 4 and 2 minutes, respectively. There are two people serving the customers at the place where the foods are...
-
4. The orbit of asteroid Vesta has a semimajor axis of 2.36 AU and an eccentricity of 0.089. Its mass is 2.59x1020 kg and, like all asteroids, it orbits the Sun, whose mass is 1.99x1030 kg. Calculate...
-
Differentiate between welding and brazing.
-
What does non-recourse financing mean?
-
Solve each problem. 290% of 137 miles is what?
-
Rank the following dienophiles in order of their expected reactivity in the Diels?Alder reaction. CH CH NC NC CN c=C CH c=c c=C c=C NC CN
-
1, 3-Cyclopentadiene is very reactive in DielsAlder cyclo addition reactions, but 1, 3-cyclohexadiene is less reactive and 1, 3-cycloheptadiene is nearly inert. Explain. (Molecular models are...
-
1, 3-Pentadiene is much more reactive in Diels?Alder reactions than 2, 4-pentadienal. Why might this be? H 1,3-Pentadiene 2,4-Pentadienal
-
Suppose the correlation between the stock euro returns of Siemens and the USD/EUR exchange rate is 0.2. The standard deviation of the USD/EUR is 10% and the standard deviation of Siemens's stock euro...
-
list and describe the three key client-related factors that the advisor is required to consider when developing a "suitable" investment portfolio for their client. Please cite resources used
-
Year 1 2 3 Amount ($) 2000 3000 4000 An investment made today will pays you the above cash flows at the end of each year. If your required rate of return is 5% annual interest, how much will you pay...

Study smarter with the SolutionInn App


