Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3
Question:
Explain which of the reaction would provide a better synthesis of3-hexanone.
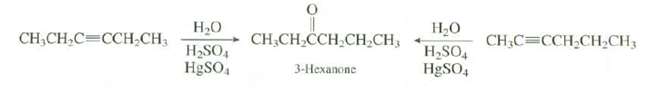
Transcribed Image Text:
Н.О Н.О CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
The left synthesis starting with 3hex...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain which route would provide a better synthesis of theseethers: CH3 CH, CH3 CHI + CH,CO a) CH.O + CH,CCI CH3 CH,COCH3 H3 CH3 CH, CH,OCHCH,CH; CH,Br CH,0 Br + CH;CHCH,CH3 b) + CH;CHCH,CH,
-
Explain which of these reactions would provide a better synthesis of2-pentene: Br CH,OH CH,CH,CHCH,CH, + CH;0 CH,CH=CHCH,CH3 Br CH, CH CH=CHCH CH, CH,CHCH,CH,CH; + CH,0
-
Explain which of the following approaches to strategy formulation is more likely to generate economic profits: (a) Evaluating external opportunities and threats and then developing resources and...
-
What is the common name for the following structure? CH3 CH3 -C-Br 1 CH3 Isobutyl bromide Osec-Butyl bromide O Neobutyl bromide O Isopropyl methyl bromide t-Butyl bromide
-
Ferguson Gas wants to move its sales order system online. Under the proposed system, gas stations and other merchants will use a secure site to check the availability and current price of various...
-
A differential leveling circuit began on BM Hydrant (elevation 1823.65 ft) and closed on BM Rock (elevation 1841.71 ft). The plus sight and minus sight distances were kept approximately equal....
-
Suppose the firm is hiring labor and capital and that the ratio of marginal products of the two inputs equals the ratio of input prices. Does this imply that the firm is maximizing profits? Why or...
-
Blooper's analysts have come up with the following revised estimates for its magnoosium mine Conduct a sensitivity analysis using the revised data. Label your answers as follows: Range Initial...
-
You plan to invest $10,000 into your new savings account the end of the year for the next 10 years. The interest earned in the account is 8% per year. How much do you expect to have in the account at...
-
Yang Inc. operates a retail operation that purchases and sells snowmobiles, among other outdoor products. The company purchases all inventory on credit and uses a periodic inventory system. The...
-
Show the products of thesereactions: 1) Hg(O,CCH,) . 2) NaBH3. NAOH 1) Hg(O,CCH,)2, H,0 2) NaBH4, NaOH b) a) . H SO4 . H2SO4 d) HgSO, H9SO,
-
Show the products of thesereactions: 1) BH3, THF 1) BH3. THF 2) .. NaOH b) 2) ,, NaOH 1) BH3, THF 2) H,O2. NaOH CH-CH3
-
Establish identity. csc 4 - csc 2 - cot 4 + cot 2
-
One noteworthy trend concerning Australia's current economic status is that "its links to Europe are eroding". What does this statement imply? What recent trends across the world are affecting...
-
Describe Empowerment, Psychological Empowerment and Community Empowerment ?
-
You are considering the following two projects and can take only one. Your cost of capital is 10.5%. The cash flows for the two projects are as follows ($ million): Project A B a. What is the IRR of...
-
Bond J has a coupon rate of 6 percent and Bond K has a coupon rate of 12 percent. Both bonds have 15 years to maturity, make semiannual payments, and have a YTM of 9 percent. a. b. If interest rates...
-
Huawei is growing steadily, and investors expect both the stock price and the dividend to increase at 8% per year. Three investors Ahmed, Badar and Khalid are interested to invest in Huawei's shares....
-
How does the Supremacy Clause of the U.S. Constitution affect the ability of the U.S. states to pass legislation?
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
Consider a solution that contains both C 5 H 5 N and C 5 H 5 NHNO 3 . Calculate the ratio [C 5 H 5 N]/[C 5 H 5 NH + ] if the solution has the following pH values: a. pH = 4.50 b. pH = 5.00 c. pH =...
-
Histamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List them in order of increasing basicity, and explain yourordering. NH2 Histamine H.
-
Oxazole is a live-membered aromatic heterocycle. Would you expect oxazole to be more basic or less basic than pyrrole?Explain. Oxazole N:
-
Protonation of an amide using strong acid occurs on oxygen rather than on nitrogen. Suggest a reason for this behavior, taking resonance intoaccount. :0: H2SO4 NH2 NH2 R.
-
You are a supervisor at your company. You've overheard Employee A making offensive comments to other employees, and Employee B has complained about these comments. You've given Employee A a verbal...
-
What is the probability of developing and dying from cancer today?
-
On January 1, 2024, Majestic Mantles leased a lathe from Equipment Leasing under a finance lease. Lease payments are made annually. Title does not transfer to the lessee and there is no purchase...

Study smarter with the SolutionInn App


