Explain which the most acidic hydrogen's in these compoundsare c) CH,CCH,CH, b) PHCH CCH3
Question:
Explain which the most acidic hydrogen's in these compoundsare
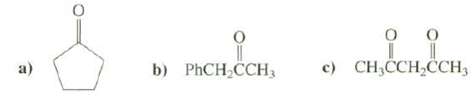
Transcribed Image Text:
c) CH,CCH,ČCH, b) PHCH CCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
a The circled hydrogens are more acidic because the conjugate base is stab...View the full answer

Answered By

Jayshree Rathi
Hello Students!
This is Jayshree Rathi. I work on a number of renowned student-centric channels such as Chegg, coursehero, as a certified private tutor.
If you are looking for relevant and original content to complete your assignments, essays, and homework, then contact me and within the promised time, I will deliver you your personalized academic work and help you score the best.
4.80+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which is the most acidic hydrogen in CH3CH2C = CH?
-
Which is the most acidic hydrogen in each of these compounds? (a) H 2 NCH 2 CH 2 OH (b) CH 3 CH 2 OH (c) CH 3 SH
-
Identify the most acidic hydrogen in each of these compounds: a) HOCCH,CH,SOH 0 CO H 0 I e) CHCCHCOCHCH3 0 b) CH-CH,CH,C=N d) 0 CHOH f) HNCHCOH
-
1. Let f(x) = 2x+6 if x < -1 3x+2 if x -1 (a) What is lim, -1- f(x)? Your answer will depend on b. (b) What is lim,-1+ f(x)? (c) For what values of b does limx-1 f(x) exist? 2. Suppose that f and g...
-
When Theo Chocolate first started is production, the company offered an exotic line of dark-chocolate and milk-chocolate bar and truckles. These early treats had unusual names such as the 3400...
-
Diane Clubb is trying to convert income statement items from an accrual basis to a cash basis. Accounts Receivable at the beginning of the year totalled $205,000. At the end of the year Accounts...
-
What is Support and Handling Equipment (CSE)? Give examples.
-
Hindi Company has the following production data for April: units transferred out 40,000, and ending work in process 5,000 units that are 100% complete for materials and 40% complete for conversion...
-
Capacitor 1 (C) has a capacitance of 1.2 F with a vacuum between the plates and the area of the plates of C, is 8.7 cm. Capacitor 2 (C2) has a plate area of 1.8 cm and a capacitance of 3.1 F when...
-
Explain the difference between a required RFC and a recommended RFC.
-
Draw the structures for these compounds: (a) (Z)-Oct-3-en-2-one (b) 3-Ethylheptanal (c) 2, 4-Pentadienal (d) 3, 4-Dimethylbenzaldehyde (e) 1-Phenyl-1-propanone (f) 2, 2, 6, 6-Ttramythleyelohexanone
-
(a) Is sodium hydroxide a strong enough base to completely remove a proton from the ?-carbon acetone; that is, does this equilibrium lie nearly completely to the right when sodium hydroxide is the...
-
The trinomial 6x 2 + 7x - 20 may be factored using substitution. Replace t with 6x in the answer from Exercise 79, and simplify to obtain the correct factorization of the trinomial. Data from in...
-
what extent do decision-making frameworks, including rational choice theory, prospect theory, and bounded rationality, accurately capture the complexities of decision-making in real-world contexts,...
-
What role does emotional intelligence play in facilitating effective decision-making, particularly in high-stakes situations where emotions may influence judgment and risk perception, and how can...
-
Determine the critical points of the function and classify it as a maximum, minimum, or chair point 1) f(x,y) =2xy-=(x+y4) +1 2) f(x,y) =-5x+4xy-y+16x+10 3) z=x-xy-y2-3x-y 4) z=ey
-
What role does decision framing play in shaping preferences and choices, and how can reframing techniques, such as perspective-taking and scenario analysis, be utilized to broaden decision-makers'...
-
Suppose individuals get utility from consumption and disutility from working according to the following utility function: (c) = -1 where c and I denote consumption and hours of work, respectively,...
-
An engine that operates on a Brayton cycle uses air as its working substance, compressing ambient air initially at a pressure of \(1.0 \mathrm{~atm}\) to \(6.0 \mathrm{~atm}\). (a) What is the...
-
Consider the following cash flows in Table P5.5. (a) Calculate the payback period for each project. (b) Determine whether it is meaningful to calculate a payback period for project D. (c) Assuming...
-
Under the European Unions Common Fisheries Policy, countries are allocated quotas for the amounts of fish that their fishermen can catch in various areas of the sea. In most European nations,...
-
Draw the following molecules in chair conformations, and tell whether the ring sub-stituents are axial orequatorial: CH (b) (a) CH
-
Lithocholic acid is an A-B cis steroid found in human bile. Draw Lithocholic acid showing chair conformations as in figure, and tell whether the hydroxyl group at C3 is axialequatorial. An A-B trans...
-
Compare the structures of lanosterol and cholesterol, and catalog the changes needed for the transformation.
-
The following information relates to production activities of Mercer Manufacturing for the year. Actual direct materials used 16,800 pounds at $4.45 per pound Actual direct labor used 17,435 hours at...
-
The bond market is the largest global financial market. Corporations, municipalities, and governments need to raise funds constantly, and debt is one of the easiest and cheapest ways to do it. Equity...
-
Elbow Bend Hotel Rooms Information Total Rooms=100 Deluxe room=40 Executive rooms= 50 Suites= 10 Suites are 100% sold every month and the other two categories split the rest of the rooms sold...

Study smarter with the SolutionInn App


