Identify the most acidic hydrogen in each of these compounds: a) HOCCH,CH,SOH 0 CO H 0
Question:
Identify the most acidic hydrogen in each of these compounds:
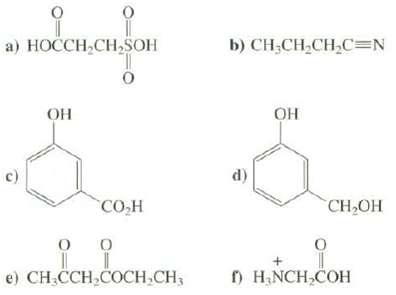
Transcribed Image Text:
요 a) HOCCH,CH,SOH ОН 0 CO H 0 I e) CH₂CCH₂COCH₂CH3 0 b) CH-CH,CH,C=N d) ОН 0 CH₂OH f) H₂NCH₂COH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a A sulfonic acid is a stronger acid than a carboxylic acid HOCCHCHSO TH b The circled hydrogen is ...View the full answer

Answered By

Payal Mittal
I specialize in finance and accounts.You can ask any question related to til undergradution.Organizational behaviour and HRM are my favourites for you can always relate to them and is an art with practical knowledge base.
4.90+
226+ Reviews
778+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which is the most acidic hydrogen in each of these compounds? (a) H 2 NCH 2 CH 2 OH (b) CH 3 CH 2 OH (c) CH 3 SH
-
Identify the most acidic proton in each of the following compounds: Xo Xx
-
Identify the most acidic proton in each of the following compounds and explain your choice: (a) (b) 0-H F3C 0-H CI. CI
-
What responsibilities can a Crew Boss delegate to a subordinate supervisor? (Select all that apply) Re-supplying crew and equipment Communicating crew wake up time for next operational period...
-
Apollo Can Ltd manufactures recyclable soft drink cans. A unit of production is a box of 12 cans. The following standards have been set by the production engineering staff and the factory accountant:...
-
Explain whether each of the following events would cause the nations currency to become overvalued or undervalued relative to its implied purchasing power parity value: (a) Russia becomes a leading...
-
An airplane flies at \(150 \mathrm{~km} / \mathrm{hr}\). (a) The airplane is towing a banner that is \(b=0.8 \mathrm{~m}\) tall and \(\ell=25 \mathrm{~m}\) long. If the drag coefficient based on area...
-
New equipment purchase, income taxes. Innovation, Inc. is considering the purchase of a new industrial electric motor to improve efficiency at its Fremont plant The motor has an estimated useful life...
-
Fanshawe college Course Code : MGMT 6147 : Trends in Management & Leadership Module & Case: #10: Motivated reasoning, leadership, and team performance. 1) Please provide a brief summary of the case....
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Complete these equilibrium reaction sin the most reasonable manner possible using the curved arrow convention to show the movement of electrons in the reactions, Predict whether the reactants or the...
-
Show the products of these acid-base reactions and predict whether the equilibria favor the reactants or the products: a) CHCCHCCHCH + OCHCH b) CHCHNO + CHO: CH3 (c) CH3COCH, + CHCH- 10 1:Z: LL CH,...
-
Suppose that the required reserve ratio is 9%, currency in circulation is \($620\) billion, the amount of checkable deposits is \($950\) billion, and excess reserves are \($15\) billion. a. Calculate...
-
Discuss the following concepts: preservation of life with limits, euthanasia, advance directives, futility of treatment, withholding and withdrawal of treatment, and do-not-resuscitate orders.
-
Describe the responsibilities of patients as reviewed in this chapter.
-
Describe the various roles of advanced practice nurses.
-
Describe advances in medicine that have contributed to improving the quality of patient care.
-
Describe the rights of patients as reviewed in this chapter.
-
Graph the three-month Treasury bill interest rate, the ten-year government bond interest rate, and the CPI inflation rate (based on the percentage change in the CPI from one year earlier) on the same...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Prove the identity. sin( x) = sin x
-
Compound A, MW = 86, shows an IR absorption at 1730 cm1 and a very simple 1H NMR spectrum with peaks at 9.7 (1 H, singlet) and 1.2 (9 H, singlet). Propose a structure for A.
-
Compound B is isomeric with A (Problem 19.63) and shows an IR peak at 1715 cm1. The 1H NMR spectrum of B has peaks at 2.4 (1 H, septet, J = 7 Hz), 2.1 (3 H, singlet), and 1.2 (6 H, doublet, J =...
-
The 1 H NMR spectrum shown is that of a compound with formula C 9 H 10 O. How many double bonds and/or rings does this compound contain? If the unknown has an IR absorption at 1690 cm ?1 , what is a...
-
Do you envision the business as starting by targeting specific needs of a single target community (e.g. the target is San Francisco), targeting specific needs of a similar set of communities (e.g....
-
Define social media. ( 20 points ) How has social media changed the way marketers and consumers communicate with one another? Explain the implications for marketers. Describe how marketing managers...
-
Find an article or video offering advice on choosing the best financing option for a business. Answer the following questions: Which types of financing did they discuss? What advice did they offer?...

Study smarter with the SolutionInn App


