Explain why one of these compounds reacts readily by an E2 mechanism when treated with sodium ethoxide
Question:
Explain why one of these compounds reacts readily by an E2 mechanism when treated with sodium ethoxide in ethanol but the other doesnot:
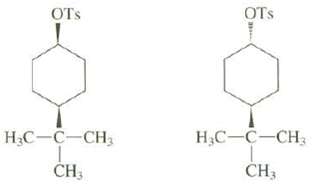
Transcribed Image Text:
OTs OTS Н.С— С—СH, Н.С—С—СH, CH; CH3 зн
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
The bulky tBu group must be equatorial and locks the ring in a single conformation For e...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
When cis-1-bromo-4-tert-butylcyclohexane is treated with sodium ethoxide in ethanol, it reacts rapidly; the product is 4-tert-butylcyclohexene. Under the same conditions, trans-...
-
The reaction between 2-bromobutane and sodium ethoxide in ethanol gives rise to three E2 products. What are they? Predict their relative amounts.
-
The reaction in eq. 7.17 occurs by an E2 mechanism (review eqs. 7.22 and 7.23). By what mechanism does the reaction in eq. 8.8 occur? H,sO CH3CHOH + HOCH2CH3 140%. CH,CHOC ethanol diethyl ether
-
Mike has the following monthly information: Salary $4,000 Rent 1,800 700 Car payment Investment Income 200 Meals 900 Groceries 700 Student Loan Payment 400 Other Expenses 600 1. What is Mike's...
-
Buzzsaw Manufacturing incurred $267 200 of manufacturing overhead costs during the past year. However, only $232 000 of overhead was applied to production. At the conclusion of the year, the...
-
Harry has been arrested because of a parole violation warrant, the result of being charged with a felony domestic assault in which he busted his girlfriend's lip. Harry hires a private attorney who...
-
If we have a model for the short rate \(r(t)\), show that (a) the zero coupon bond price can be calculated as: \[P(t, s)=\mathbf{E}_{t}\left[e^{\int_{t}^{s} r(u) d u}...
-
The required steps in the accounting cycle are listed in random order below. List the steps in proper sequence. (a) Prepare a post-closing trial balance. (b) Prepare an adjusted trial balance. (c)...
-
In what ways do organizations navigate the tension between fostering innovation and maintaining operational stability during periods of transformative change ? Explain
-
GRW Company has the following account balances at the end of the year, before adjusting and closing entries. (All numbers are in thousands of dollars.) Required: Calculate and interpret the year-end...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
Show the products of these elimination reactions and indicate which ismajor: OTs . b) CH,OH + CH,0 + OH ELOH CI E:OH + CH,CH,O c)
-
What determines the quality of earnings?
-
Electrostatic work \(W\) is done on a charged particle as it travels \(10 \mathrm{~mm}\) along a straight path from point A to point \(B\) in an electric field. If you return the particle to point...
-
What is the electric field magnitude a radial distance \(r\) from the central length axis of an infinitely long thin rod carrying a positive charge per unit length \(\lambda\) ?
-
What is the electric field a distance \(d\) from a thin, infinite nonconducting sheet with a uniform positive surface charge density \(\sigma\) ?
-
An electron moves from point \(A\) to point \(B\) under the influence of an electrostatic field in which the potential difference between A and B is negative. (a) If your system includes both the...
-
Express the SI unit of electric field in terms of joules and other SI base units.
-
Repeat Problem 7.3 using methyl acetate as solvent, for which \(K_{D}=1.273 \mathrm{~kg}\) water/kg methyl acetate. Data From Problem 7.3:- A feed of \(13,500 \mathrm{~kg} / \mathrm{h}\) consists of...
-
Use this circle graph to answer following Exercises. 1. What fraction of areas maintained by the National Park Service are designated as National Recreation Areas? 2. What fraction of areas...
-
Simplify each expression. n+ 5+2 +5n
-
At what approximate positions might the following compounds show IRabsorptions? (c) (b) CH CH (a) CH3CH2CH3 CHCCH2CH3CH2 CHCH2C3CH (f) . (d) (e) " CCH2CH2COCH CH
-
Assume you are carrying out the dehydration of 1-methylcyclohexanol to yield 1-methykyclohexene. How could you use infrared spectroscopy to determine when the reaction is complete?
-
Assume that you are carrying out the base-induced de-hydro bromination of 3-bromo-3-methylpentane (Section 11.7) to yield an alkene. How could you use IR spectroscopy to tell which of two possible...
-
Problem: Module 3 Textbook Problem 5 Learning Objective: 3-6 Using the straight-line method show how bonds issued at a discount. affect financial statements Diaz Company issued $91,000 face value of...
-
Manvir had to make payments of $1,125 every 6 months to settle a $22,000 loan that he received at 4.52% compounded semi-annually. a. How long did it take to settle the loan?
-
If I invest a single amount of $14,000 in an account earning 8% p.a. compounding quarterly for 5 years, how much interest will I have earned in those 5 years?

Study smarter with the SolutionInn App


