Figure shows a slab with dimensions d1 = 11.0 cm, d2 = 2.80 cm, and d3 =
Question:
Figure shows a slab with dimensions d1 = 11.0 cm, d2 = 2.80 cm, and d3 = 13.0 cm. Half the slab consists of aluminum (density = 2.70 glcm3) and half consists of iron (density = 7.85 g/cm3).What are(a) The x coordinates,(b) The y coordinate, and(c) The e coordinates of the slab's center ofmass?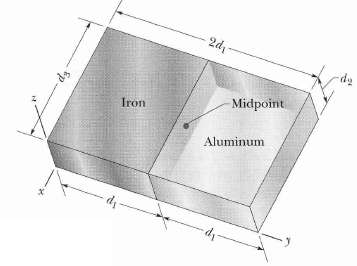
Transcribed Image Text:
2d - Midpoint Iron Aluminum di -
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
We use Eq 95 to locate the coordinates a By symmetry Xcom d...View the full answer

Answered By

ATUL GAUTAM
• Year 2018, Class 10 from Ryan International School - secured 94.2%
• Year 2020, Class 12 (Commerce + maths) from Ryan International School - secured 91.6%
• Bachelor of Commerce Pursuing from MJP Rohilkhand University (Swami Shukdevanand College) - secured
1St division (1st & 2nd year)
Current year – 3rd year
• CA Pursuing from Institute of Chartered Accountant of India (ICAI) - secured Foundation – 238/400
Inter – Pursuing
• Expert user of MS DOS and MS Office (Word, Excel, Power Point)
• 1 year of teaching experience in offline mode in the field of accounts and maths to ca foundation students.
• Currently working in different online platform as Q/A Expert.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Mechanics questions
-
The figure shows a composite slab with dimensions d = 11.0 cm, d = 2.76 cm, and d3 = 12.9 cm. Half the slab consists of aluminum (density = 2.70 g/cm) and half consists of iron (density = 7.85 g/cm)....
-
Assume that your utility function is equal to U = [E(r)] - Ao. You create a portfolio of one risky stock with the expected return of and standard deviation of p, and one risk-free stock with the...
-
A centrifugal pump has d1 = 7 in, d2 = 13 in, b1 = 4 in, b2 = 3 in, β1 = 25°, and β2 = 40° and rotates at 1160 r/min. If the fluid is gasoline at 20°C and the flow enters the...
-
How do you identify the potential classes in a problem domain description?
-
Explain how the Financial Reform Act of 2010 and the rules issued to implement it attempted to reduce the risk in the financial system resulting from the use of credit default swaps.
-
Three components are connected to form a system as shown in the accompanying diagram. Because the components in the 2-3 subsystem are connected in parallel, that subsystem will function if at least...
-
A game of chance based on a spinning wheel is available that pays \(n\) times money bet in the case of a win and nothing in the case of a loss. A gambler has developed a device by which he may...
-
Cran Health Products is a cranberry cooperative that operates two divisions, a harvesting division and a processing division. Currently, all of harvestings output is converted into cranberry juice by...
-
Find the change in a disk's angular speed (in rad/s) if its mass and radius are 0.230 kg and 0.180 m, respectively
-
The following transactions occurred during the month for Teresa Parker, CPA: Jun. 1 Parker opened an accounting firm by contributing $13,200 cash and office furniture with a fair market value of...
-
Figure shows a cubical box that has been constructed from uniform metal plate of negligible thickness. The box is open at the top and has edge length L = 40 cm. Find(a) The x coordinates,(b) The y...
-
A uniform soda can of mass 0.140 kg is 12.0 cm tall and filled with 1.3l kg of soda (Figure).Then small holes are drilled in the top and bottom (with negligible loss of metal) to drain the soda. What...
-
Generally aldehydes are more susceptible to oxidation in air than are ketones. Use acetaldehyde and acetone as examples and show why ketones such as acetone are more stable than aldehydes in this...
-
From June to September 2022, oil prices in the United States declined by about 35 percent. During these same months, U.S. oil production remained roughly constant, and stocks of oil in storage...
-
In 2018, an unusual situation developed with respect to production of natural gas in the Permian Basin of western Texas and New Mexico. A news article noted the following two facts: i. Shale drillers...
-
Juan Perez argues that Learning how to do business analysis and valuation using financial statements is not very useful, unless you are interested in becoming a financial analyst. Comment.
-
Should the capital budgeting committee accept the internal auditors first and second suggestions, respectively? A. No for Suggestions 1 and 2. B. No for Suggestion 1 and Yes for Suggestion 2. C. Yes...
-
Agricultural and industrial activity have substantially increased the reactive nitrogen content of the atmosphere. To investigate the potential consequences on ecosystems, Stevens et al. (2015)...
-
If the two securities are uncorrelated, the expected standard deviation of an equalweighted portfolio is closest to: A. 14.00%. B. 14.14%. C. 20.00%. Use the following data to answer Question. A...
-
The slopes of the tangents at the points where the curve y = x2 - 4x intersects the X-axis is 1) 1 2) +2 3) +3 4) +4
-
(a) Use of hydrogen fusion to supply energy is a dream that may be realized in the next century. Fusion would be a relatively clean and almost limitless supply of energy, as can be seen from Table...
-
The compression ratio of an Otto cycle, as shown in Figure 22.13, is VA/VB = 8.00. At the beginning A of the compression process, 500 cm3 of gas is at 100 kPa and 20.0C. At the beginning of the...
-
An ice tray contains 500 g of liquid water at 0C. Calculate the change in entropy of the water as it freezes slowly and completely at 0C.
-
At a pressure of 1 atm, liquid helium boils at 4.20 K. The latent heat of vaporization is 20.5 kJ/kg. Determine the entropy change (per kilogram) of the helium resulting from vaporization
-
Given the matrix A -3 0 -14 3 4 6 9115 7701 a) Determine all solutions of the homogeneous system Ax = 0. b) Determine if the columns of A span R.
-
Summarize each data source and include them for substance abuse and alcohol in the military. Analyze each data source for substance abuse and alcohol in the military for trustworthiness and accuracy....
-
On January 1, 2023, Holland Corporation paid $7 per share to a group of Zeeland Corporation shareholders to acquire 60,000 shares of Zeeland's outstanding voting stock, representing a 60 percent...

Study smarter with the SolutionInn App


