For a given a hydrogen atom to be acidic, the C?H bond must be parallel to the
Question:
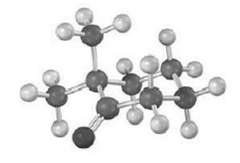
For a given a hydrogen atom to be acidic, the C?H bond must be parallel to the p orbital?s of the C=O bond (that is, perpendicular to the plane of the adjacent carbonyl group). Identify the most acidic hydrogen atom in the conformation shown for the following structure. Is it axial or equatorial?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
HC CH3 H Enolization can occur on only one side of the car...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The relative orbital levels for the hydrogen atom can be represented as Draw the relative orbital energy levels for atoms with more than one electron, and explain your answer. Also explain how the...
-
A force P is applied to the lever of an arbor press. Knowing that P lies in a plane parallel to the yz plane and that 20 My = − N⋅m and Mz = −3.5 N⋅m, determine the moment Mx of...
-
A hydrogen atom in a particular orbital angular momentum state is found to have j quantum numbers 7/2 and 9/2. What is the letter that labels the value of l for the state?
-
Calculate investment balance four years after acquisition Ray Corporation owns a 40 percent interest in the outstanding common stock of Ton Corporation, having acquired its interest for $2,400,000 on...
-
What are the four parts of successful persuasive messages?
-
Suppose that only 80% of the STUDENT records from Exercise 17.28 have a value for PHONE, 85% for MAJORDEPTCODE, 15% for MINORDEPTCODE, and 90% for DEGREEPROGRAM, and we use a variable-length record...
-
In a certain examination, the mean score per student for 20 students is 75 with a standard deviation of 8.5. Using Chebychevs Theorem, determine at least how many of the students scored between 58...
-
A quant jock from your firm used a linear demand specification to estimate the demand for its product and sent you a hard copy of the results. Unfortunately, some entries are missing because the...
-
1. Create database named 'Watch'. Create a table named 'Watch' with the data fields as stated below in the database. Watch ID (Primary Key) Model Kind of watch glass Water resistance Strap Validation...
-
In problem 0-4-3[BEZ], assume the energetic efficiency (work transfer to the elevator (desired) to the electrical work transfer (required) to the motor), , of the system to be 80%, (a) Determine the...
-
Unlike most ?-dike tones, the following ?-dike tone has no detectable enol content and is about as acidic as acetone. Explain.
-
Identify all the acidic hydrogen?s (pKa (b) (c) (a) CHCH2H3 CH H-CH23H C02CH (f) COCI (d) (e) CHCH2CC 3CH2 CH CH2CN
-
Describe how financial leverage can increase return on owners equity.
-
What are the advantages of using the T7 polymerase to control expression ?
-
Discuss the importance of signing in with a Microsoft Account or Local Account in Windows 10.?
-
What effect did the UV exposure have on the control plasmid DNA? Explain what evidence supports this conclusion.
-
7. A shouldered screw contains three hollow right circular parts on the screw before a nuts is tightened against the shouldered. To sustain the function, the gap co must equal or exceed 0.08mm. The...
-
Jack and Jennifer have been married for 20 years and always file a joint return, but never itemize their deductions. Jack and Jennifer have gross income of $80,000 and deductions for adjusted gross...
-
A national survey asked people, How often do you have dinner outside instead of at home? The frequencies were as follows. (a) Construct a relative frequency distribution. (b) What percentage of...
-
Which of the following gives the range of y = 4 - 2 -x ? (A) (- , ) (B) (- , 4) (C) [- 4, ) (D) (- , 4] (E) All reals
-
How many sp 3 hybrid orbitals result from the hybridization of one s and three p orbitals? (a) 1 (b) 2 (c) 3 (d) 4
-
Characterize each step of the mechanism in Eq. 18.42b in terms of the fundamental processes discussed in the previous section. Give the electron count and the oxidation state of the metal in each...
-
What two sets of aryl bromide and alkene starting materials would give the following compound as the product of a Heck reaction? C'
-
What product is expected when cyclopentene reacts with iodobenzene in the presence of tri-ethlamine and a Pd(0) catalyst?
-
Compare and contrast different deadlock prevention techniques such as resource ordering, the "hold and wait" condition, and preemptive resource allocation. Which methods are most effective for...
-
The following information is taken from Aden Company's records: Product Group Units Cost/Unit Market/Unit A 1 700 $1.10 $0.90 B 1 250 1.50 1.55 C 2 150 4.90 5.15 D 2 100 6.50 6.40 E 3 80 25.00 24.60...
-
American Food Services, Incorporated leased a packaging machine from Barton and Barton Corporation. Barton and Barton completed construction of the machine on January 1, 2024. The lease agreement for...

Study smarter with the SolutionInn App


