Identify all the acidic hydrogen?s (pKa (b) (c) (a) CHCH2H3 CH H-CH23H C02CH (f) COCI (d) (e)
Question:
Identify all the acidic hydrogen?s (pKa
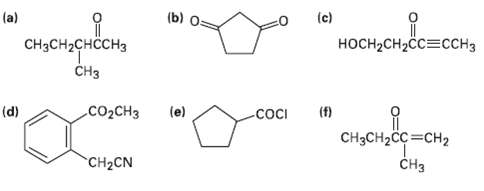
Transcribed Image Text:
(b) (c) (a) CHзCH2снссH3 CHз носH-CH2сс3ссHз C02CHЗ (f) COCI (d) (e) CHзCH2CC 3CH2 CHз CH2CN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
Acidic hydrogens are bold The most acidic hydrogens are the t...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Hydrogen cyanide (HCN) has a pKa of 9.1. What is its Ka? Is HCN a strong or a weak acid?
-
Identify which of the following compounds is more acidic and explain your choice.
-
Identify the more acidic compound in each of the following pairs: (a) CF3CH2CO2H or CF3CH2CH2CO2H (b) CH3CH2CH2CO2H or CH3CPCCO2H (c) (d) (e) (f) (g) CO2H CO2H or CO,H CO2H o F- COH CO.H -CO-H or...
-
A general ledger trial balance at June 30, 2011, for Millar City is as follows: Millar City uses a purchases basis in accounting for supplies. Open encumbrances are considered constrained by the...
-
How are direct-mail sales messages and e-mail sales messages similar, and how are they different?
-
Use the following formulas to return the t-scores you need in the above formula from Excel. =t.inv(probability, degrees of freedom) To retrieve the lower t (t. part of formula): probability = a/2 and...
-
Explain the relationship between classifier systems and production systems. How are classifier systems built? What advantages do they have over production systems?
-
Dude Corporation manufactures specialty lines of mens apparel. During February, the company worked on three special orders: B-2, B-3, and B-4. Cost and production data for each order are as follows....
-
Part of the Federal Agenda for Criminal Justice Reform proposed by members of Congress contains a proposal to incentivize states to reduce their prison populations. Enact the Reverse Mass...
-
What limits or controls do you believe should be in place for children of varying ages?
-
For a given a hydrogen atom to be acidic, the C?H bond must be parallel to the p orbital?s of the C=O bond (that is, perpendicular to the plane of the adjacent carbonyl group). Identify the most...
-
Rank the following compounds in order of increasingacidity: (a) CH3CH2CO2H (b) CH3CH2OH (c) (CH3CH2)2NH (d) CH3COCH3 (e) (f) CCI3CO2H CCH-CCH3
-
Magma is generated at divergent and convergent plate boundaries. What type of magma is dominant at each boundary? Why are they different?
-
At a bake sale, 15 items were sold for a total of $31. cobblers sold for $3.50 each, strudels sold for $2.00 each, and cookies sold for $1.00 each. The number of cookies was 25 less twice the number...
-
Beyonce owns a small business that manufactures and sells lamps. The lamps cost $7.50 each to manufacture, plus a monthly overhead of $3600. She sells the lamps for $30 each. a) Set up a system of...
-
Oaktree, Inc. is a merchandiser of inlaid wooden boxes. The company sold 7,500 units during the year. The company has provided the following information: Sales Revenue Purchases (excluding freight...
-
Convert the following branch and jump instructions to Machine Code. Assume fact is at byte address 256 6Y fact: addi $ap, $ap, -8 beq $50, $to, L1 sw Sa0, 0($sp) slti $to, $a0, 12 addi $v0, $zero, 11...
-
22. Complete the table below to describe why different types of insurance are relevant to an Allied health Workplace Type of Business insurance Description Workers Compensation Public liability
-
(a) Describe the three parts of a passive continental margin. (b) Describe the parts of an active continental margin.
-
For the following exercises, write the polynomial function that models the given situation. Consider the same rectangle of the preceding problem. Squares of 2x by 2x units are cut out of each corner....
-
Use MO theory to predict the bond order in H 2 . Is the H 2 bond a stronger or weaker bond than the H 2 bond?
-
Complete the following Pd(0)-catalyzed Suzuki reactions by giving the coupling product. Include the stereochemistry of the products. BOH CH3 CH3O
-
Give two different pairs of starting materials that could be used to prepare the following compound by a Suzuki coupling. CH3 Ph C-C
-
Draw structures analogous to those in Eqs. 18.59a-d for the catalytic intermediates formed in the conversion of 1, 7-octadiene to cyclohexene and ethylene catalyzed by the G2 catalyst. Eqs. 18.59a-d...
-
Schwiesow Corporation has provided the following information: Cost per Cost per Unit Period Direct materials $ 7.05 Direct labor $ 3.50 Variable manufacturing overhead $ 1.65 Fixed manufacturing...
-
Choose an industry in which you are interested in working. How is that industry trending? What internal and external factors may affect the direction of the organization? For your initial post,...
-
The DeVille Company reported pretax accounting Income on its Income statement as follows: 2024 2025 2026 2027 $ 420,000 340,000 410,000 450,000 Included in the income of 2024 was an installment sale...

Study smarter with the SolutionInn App


