For a given value of [HL]org in Equation 22-13, over what pH range (how many pH units)
Question:
For a given value of [HL]org in Equation 22-13, over what pH range (how many pH units) will D change from 0.01 to 100 if n = 2?
Equation 22-13
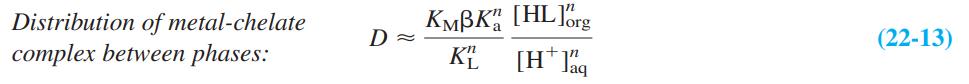
Transcribed Image Text:
Distribution of metal-chelate complex between phases: KMBK [HLErg K“ [H* ig D (22-13)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
D CH n where C K M n a HL n o...View the full answer

Answered By

John Kimutai
I seek to use my competencies gained through on the job experience and skills learned in training to carry out tasks to the satisfaction of users. I have a keen interest in always delivering excellent work
4.70+
11+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Show that the number of different electron states possible for a given value of n is 2n2.
-
(a) For a given value of the principal quantum number n, how many values of the orbital quantum number are possible? (b) For a given value of how many values of the orbital magnetic quantum number m...
-
For a given value of the principal quantum number, n, how do the energies of the s, p, d, and f sub-shells vary for? (a) Hydrogen, (b) A many-electron atom
-
Why do countries respond differently in terms of flexibility and worklife balance to what seem similar economic pressures?
-
You are provided with the following information for Barton Inc. Barton Inc. uses the periodic method of accounting for its inventory transactions. March 1 Beginning inventory 2,000 liters at a cost...
-
Conduct the following analyses for the Internet usage data given in Applied Problem 11-18 in Chapter 11. a. Find the simple correlations between the following sets of variables: Internet usage and...
-
True or False. Gathering data is all that is required for a successful trending program.
-
1. What are the costs and benefits of FDI inflows for a host country such as Argentina? 2. Will foreign firms such as Repsol make decisions in the best interest of Argentina? 3. As a Spanish manager...
-
ABC; product profitability Outerwear Inc. is concerned about the profitability of its regular gloves. Company managers are considering producing only the top-quality, fleece lined, gloves. The...
-
The vectors span R3. Pare down the set {x1, x2, x3, x4, x5} to form a basis for R3. X2 X2 = X3 = X4 = 274
-
Butanoic acid has a partition coefficient of 3.0 (favoring benzene) when distributed between water and benzene. Find the formal concentration of butanoic acid in each phase when 100 mL of 0.10 M...
-
For the extraction of Cu 2+ by dithizone in CCl 4 , KL= 1.1 10 4 , K M = 7 10 4 , K a = 3 10 - 5 , = 5 10 22 , and n = 2. (a) Calculate the distribution coefficient for extraction of 0.1 M Cu 2+...
-
When appropriate round quotas to the nearest hundredth. Suppose that Turtlestan is a small country with a population of 7,500,000 that consists of four states, A, B, C, and D. There are 150 seats in...
-
Throughout the Mystery of Banking, Rothbard talk about free banking. What is free banking? Is FRB possible under free banking?
-
Explain the relationship between genetic diversity, mutation, natural selection, and evolution
-
What are the tax consequences for Nonqualified Stock Options? Incentive Stock Options? What is Restricted Stock?
-
Outline the competition intensity of the Indian Banking Industry while highlighting the innovation, digitation that has led to virtual banking and other modes of banking. Explain the importance of...
-
1) Where would a bank customer's account data be directly stored? 2) Which of the following manages the Bank database? 3) Which of the following would prevent unauthorized access to the Bank...
-
What does the 95% represent in a 95% confidence interval?
-
Suppose that you could invest in the following projects but have only $30,000 to invest. How would you make your decision and which projects would you invest in? Project Cost $ 8,000 11,000 9,000...
-
Quartz crystal microbalance. The area of the gold electrodes on the quartz crystal microbalance at the opening of Chapter 2 is 3.3 mm 2 . One gold electrode is covered with DNA at a surface density...
-
(a) Figure 1-1 shows a peak O 3 concentration of 19 mPa in the stratosphere. Figure 1-3 shows a peak concentration of O 3 of 39 ppb at ground level at one particular location. To compare these...
-
(a) How much of the primary standard benzoic acid (FM 122.12, density = 1.27 g/mL) should you weigh out to obtain a 100.0 mM aqueous solution in a volume of 250 mL? (b) What apparent mass in air will...
-
Career pathing programs arm staff with skills and provides incentives of growth, challenge, and visibility. It presents the chance to change the way a technology team is perceived internally and...
-
In order to better understand what you are trying to communicate, does Christianity and the Way of Jesus fall under a honor society, justice society or both?
-
For HOST280, I have a significant assignment. Core Values I need to be able to define what each core value means. To define these three core values. The definition is personal so that if someone asks...

Study smarter with the SolutionInn App


