Give IUPAC name for the following compounds (reddish brown =Br): (a) (b) (c)
Question:
Give IUPAC name for the following compounds (reddish brown =Br):
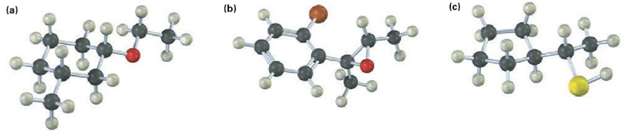
Transcribed Image Text:
(a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
H3C c cis1Ethoxy3methyl...View the full answer

Answered By

MICHAEL KICHE
I was employed studypool for the first time in tutoring. I did well since most of my students and clients got the necessary information and knowledge requested for. I always submitted the answers in time and followed the correct formatting in answering eg MLA or APA format,
Again I worked with the writers bay where I did writing and got many clients whom we worked with so closely. They enjoyed every single service I delivered to them. My answers are always correct.
4.70+
13+ Reviews
53+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give IUPAC name for the followingcompounds: (c) NC. CH 2 () (a) CHCH2CH2CH CH2 CH () CH CH2CO2H (d) (e) CHH2H2H2CH3 CHCN CH (g) Br (h) CN BRCH2CHCH2CH2CO2H
-
Give an IUPAC name for the following compounds: q, b.CH,CH2CHCH, c' .CH,HCHCH, CH3 CH,--C-CH3 CH3 ,
-
Give IUPAC name for the following compounds: (c) , (b) (a) CHCH,CHCHCH CH CH2CH2CH3 -CH CH C (e) . (f) (d) Br Br
-
The object is used to execute a static Oracle query, but the object is used to execute a dynamic Oracle query with IN and OUT parameters. a. PreparedStatement, Statement b. Statement,...
-
How do you feel about having your fingerprints, facial features, and perhaps more of your biometric features encoded in documents like your passport? Explain your answer.
-
Using Table E.6, determine the lower- and upper-tail critical values for the Wilcoxon rank sum test statistic, T1, in each of the following two-tail tests: a. a = 0.10, n1 = 6, n2 = 8 b. a = 0.05, n1...
-
What factors should be considered in setting alpha risk? How is this risk controlled?
-
Sid Patel owns a service station and has the opportunity to purchase a car-wash machine for $15,000. After carefully studying projected costs and revenues, Patel estimates that the car-wash machine...
-
Using those segments, how does the Spain Wine Market differentiate from the United States Wine Market?
-
Dorchester, Ltd. is an old-line confectioner specializing in high-quality chocolates. Through its facilities in the United Kingdom, Dorchester manufactures candies that it sells throughout Western...
-
The 1H NMR spectrum shown is that of an ether with the formula C4H8O. Propose astructure. TMS 10 9. 8. 7. 6. O ppm Chemical shift (8) Intensity 3.
-
Show the product, including stereochemistry that would result from reaction of the following epoxide with HBr.
-
A normal distribution has a mean equal to 10. What is the standard deviation of this normal distribution if the cutoff for the bottom 5% is x = 12.47?
-
1 Find the compound amount and compound interest if $4000 is invested for eight years and interest is compounded continuously at the annual rate 6%. The compound amount will be $ (Round to the...
-
In 1929, U.S. GDP was, adjusted for inflation, $1,056.6 billion. The following year it was, also adjusted for inflation, $966.7 billion. What was the growth rate of U.S. in 1930? (Do not type the...
-
The parents stay for a while and play with their child and the other children and seem happy to leave. They say goodbye and their child cries initially when they leave and needs comforting. You sit...
-
What role do telomeres play in maintaining chromosome stability, and how do they relate to cellular aging and cancer development? Explain
-
(a) Demand and supply of fuel in Namibia depends on the current price. Namibian fuel dealers determine their future prices on the basis of current price and excess demand. If excess demand for fuel...
-
The trial balance columns of the worksheet for Barbosa Apparel at June 30, 2025, are as follows. Other data: Operating expenses incurred on account, but not yet recorded, total R$1,640. Instructions...
-
Listed below are common types of current liabilities, contingencies, and commitments: a. Accounts payable b. Bank loans and commercial paper c. Notes payable d. Dividends payable e. Sales and excise...
-
Redo Problem 4.17 using Aspen Plus. Problem 4.17 Hydrogen has an auto-ignition temperature of 853 K; that is, hydrogen will ignite spontaneously at that temperature if exposed to oxygen. Hydrogen is...
-
Explain how UV spectroscopy could be used to distinguish between thesecompounds. and a) b) and c) and HO and d)
-
Butane and acetone both have a molecular mass of 58, calculate the exact masses of these compounds and explain whether they can be distinguished by high-resolution mass spectrometry.
-
What are the predicted intensities of the M + 1 peaks in the mass spectra of butane and acetone, relative to the intensity of the M+ peak? Do you think that these intensities could be used to...
-
sked byChina699 BEMIDJI STATE UNIVERSITY Department of Technology, Art & Design TADT 3217 : Materials Science & Metallurgy Hardness Testing [A continued look at the Heat Treatment of Steel] ...
-
1. Advocate Aurora Sheboygan Memorial Hospital health care product or service to be marketed in your community. 2. Conduct appropriate market research in your community to determine the demographics...
-
How do ethical leaders integrate ethical considerations into strategic decision-making processes, balancing short-term business objectives with long-term ethical imperatives to ensure sustainable...

Study smarter with the SolutionInn App


