The 1H NMR spectrum shown is that of an ether with the formula C4H8O. Propose astructure. TMS
Question:
The 1H NMR spectrum shown is that of an ether with the formula C4H8O. Propose astructure.
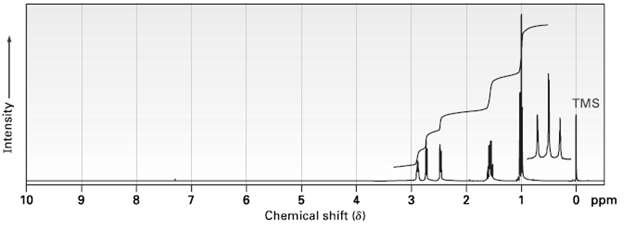
Transcribed Image Text:
TMS 10 9. 8. 7. 6. O ppm Chemical shift (8) Intensity 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
CH3CHCCH H 12Epox...View the full answer

Answered By

Rohith Bellamkonda
I am studying in IIT Indore,the most prestigious institute of India.I love solving maths and enjoy coding
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H NMR spectrum shown is that of 3-methyl-3-buten-l-ol. Assign all the observed resonance peaks to specific protons, and account for the splittingpatterns. TMS H H2H20 10 8. O ppm Chemical shift...
-
The 1 H NMR spectrum shown is that of a compound with formula C 9 H 10 O. How many double bonds and/or rings does this compound contain? If the unknown has an IR absorption at 1690 cm ?1 , what is a...
-
The 1 H NMR spectrum shown is that of a compound isomeric with the one in Problem 19.65. This isomer has an IR absorption at 1730 cm ?1 . Propose a structure. TMS 10 O ppm 1. Chemical shift (8)...
-
Provide a brief description of the seven basic steps to use JDBC.
-
Would you feel the same way about having biometric information on your driver's license as on your passport? Why or why not?
-
Calculating the motion of individual atoms in the vibrational modes of molecules (called normal modes) is an advanced topic. Given the normal modes shown in the following figure, decide which of the...
-
Ronda Rousey Fight Times Perhaps the most popular fighter since the turn of the decade, Ronda Rousey is famous for defeating her opponents quickly. The five number summary for the times of her first...
-
You were recently hired as a manager for international operations of Despoyle Chemical Corporation, a major manufacturer of dyes, fertilizers, and other industrial chemicals. Despoyle has chemical...
-
Using the BCG Matrix In the following mini-case study, you are required to categorize the corporation's business portfolio into the four boxes of the BCG (Boston Consulting Group) model. You then...
-
Six ovens were rented on December 31, with $20,000 charged to rent expense. The lease runs for 6 years with an implicit interest rate of 5%. At the end of the 6 years, Peyton will own them. Make any...
-
2-Butene-1-thiol is one component of shunk spray. How would you synthesize this substance from methyl 2-butenoate? From 1,3-butadiene CH3CH=CHCOCH3 CH3CH=CHCH2SH 2-Butene-1-thiol Methyl 2-butenoate
-
Give IUPAC name for the following compounds (reddish brown =Br): (a) (b) (c)
-
Consider two occupations (A and B) in which people have the same skills and abilities. When employed, workers in the two occupations work the same number of hours per day. In occupation A, employment...
-
For a business to achieve its objectives, there is a need for Financial Analysis and Planning. With the support of practical examples justify this statement.
-
Early Career Critical Thinking The Internal Audit department at Pluto - Cola assigns its team members to special working groups every year. Each group is assigned specific types of internal projects,...
-
1. Discuss the Electronic payment systems.
-
Explain the E-commerce payment system.
-
Write an Article Research Paper on Payment System.
-
Today many security personnel are private police, yet Fourth Amendment protection has been extended only to those searches conducted by government officials. What arguments can be made for and...
-
Write out the formula for the total costs of carrying and ordering inventory, and then use the formula to derive the EOQ model. Andria Mullins, financial manager of Webster Electronics, has been...
-
One kilogram of saturated liquid methane at 160 K is placed in an adiabatic pistion-and-cylinder device, and the piston will be moved slowly and reversibly until 25 percent of the liquid has...
-
Trans-1-Phenyl-1, 3-butadiene has max = 280 ( = 27,000) calculate the concentration of a solution that has A = 0.643 at 280nm in a 1 cm cell.
-
Nitro methane max = 275nm ( = 1.5) what kind of transition is responsible for this absorption?
-
3-Buten-2-one has max =213nm ( = 7080) and max = 320nm ( = 21) what kind of transition is responsible for each of these absorptions?
-
Determine the exact solution of the system given below using addition or subtraction of matrices or scalar multiplication 5x - ey + 3x+2y- z = -2 . = 1 0 4x-7y+ 5z =
-
We need to be able to describe systems to assist with development, to assist with use, and to assist with maintenance. To that end, diagrams provide a concise, semi-formal way of describing a system....
-
Determine when the parametric curve x = 31261+2 and y = 4t - is concave up.

Study smarter with the SolutionInn App


