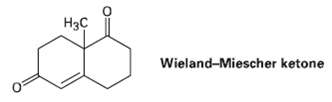
The so-called Wieland?Miescher ketone is a valuable starting material used in the synthesis of steroid hormones. How
Question:
The so-called Wieland?Miescher ketone is a valuable starting material used in the synthesis of steroid hormones. How might you prepare it from 1, 3-cyclohexanedione?
Transcribed Image Text:
Нас Wieland-Miescher ketone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
S 1 NatOEt H3C EtOH S 2 CHI 1 Na OEt EtOH 2 HCCHCO...View the full answer

Answered By

Ajeet Singh
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life.
I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge.
I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields.
Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a teacher. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
4.90+
7+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How might you prepare the following compounds using an alkylation reaction as the keystep? (b) -H CH-CH (a) CH2CH=CH2 (c) CHCH,CH2CHCEN C (d) (f) (e) CH - CH CHCHCHCH CH-CH - Hg, CH(CH3)2
-
How might you prepare Pentylamine from the following starting materials? (a) Pentanamide (b) Pentanenitrile (c) 1-Butene (d) Hexanamide (e) 1 -Butanol (f) 5-Decene (g) Pentanoic acid
-
How might you prepare 2-phenylethanol from benzyl bromide? More than one steps inneeded. CH2Br CH2CH2OH
-
Julia Robertson is a senior at Tech, and she's investigating different ways to finance her final year at school. She is considering leasing a food booth outside the Tech stadium at home football...
-
Which of the following circumstances are likely to create early-mover advantages? a) Maxwell House introduces the first freeze-dried coffee. b) A consortium of U.S. firms introduces the first...
-
(a) For n = 4, what are the possible values of l? (b) For I = 2, what are the possible values of ml? (c) If ml is 2, what are the possible values for l?
-
What are three ways that a company can identify and keep up with emerging strategic technologies?
-
The following two events occurred for Trey Co. on October 31, 2013, the end of its fiscal year. a. Trey rents a building from its owner for $2,800 per month. By prearrangement, the company delayed...
-
What are the primary sources of resistance to organizational change, and which strategies are most effective in mitigating this resistance within a complex organizational structure ?
-
Yardworx reconciled its book balance of Cash with its bank statement balance on April 30 and showed two cheques outstanding at that time, #1771 for $15,463.10 and #1780 for $955.65. The following...
-
How might the following compounds be prepared using Michael reactions? Show (he nucleophilic donor and the electrophilic acceptor in eachcase. ", , (b) . (a) CHCCH-CH2CH2CH3 CH2H2CCgHs o2Et NO2 (d)...
-
The following reactions are unlikely to provide the indicated product in high yield. What is wrong witheach? . , (a) Na* "OEt CHH2CH CHCH CHCH Ethanol (b) .oon CH2CH2CCH3 Na* "OEt + H3 Ethanol "H...
-
Draw and name all monobromo derivatives of pentane, C5H11Br.
-
Differentiate between a scalar value and a vector value.
-
Solve the following functions, 2.1) fi(n) = nlogn f2(n) = 3nloggn 2.2) fi(n) = logn f2(n) = 3loggn 2.3) f(n) =nk f2(n)=c" (k 1 and c> 1) 2.4) fi(n) =n+5n.5 f2(n) =3n2+5n+1
-
What methodologies and tools are commonly used to test and validate an RTOS? How can you ensure that the system meets real-time requirements under various conditions?
-
Distance, km 40- 30- 20- 10- 0. 0 Time, hours 1. During which time intervals is the object stationary? 2. During which time interval is the object accelerating (or decelerating) the fastest? 3....
-
When a claim related to a major car accident is evaluated, a clerk first retrieves the corresponding car accident report in the Police Reports database. If the report is retrieved, it is attached to...
-
A record player turntable initially rotating at \(33 \frac{1}{3} \mathrm{rev} / \mathrm{min}\) is braked to a stop at a constant rotational acceleration. The turntable has a rotational inertia of...
-
CLASS PERIO Solving Linear Equations: Variable on Both Sides Solve each equation. 1) 6r+ 7 = 13 + 7r 3) -7x-3x+2=-8x-8 5)-14 +66+7-26=1+5b 7) n-3n = 14-4n 2) 13-4x=1-x 4)-8-x= x - 4x 6)n+2=-14-n 8)...
-
Write the Lewis structure for each molecule or ion. a. H3COH b. OH- c. Bro- d. 0-
-
Predict the products formed by sodium hydroxide-promoted dehydrohalogenation of the following compounds. In each case, predict which will be the major product. (a) 1-bromobutane (b) 2-chlorobutane...
-
What halides would undergo dehydrohalogenation to give the following pure alkenes? (a) Hex-1-ene (b) Isobutylene (c) Pent-2-ene (d) Methylenecyclohexane (e) 4-methylcyclohexene
-
In the dehydrohalogenation of alkyl halides, a strong base such as tert-butoxide usually gives the best results via the E2 mechanism. (a) Explain why a strong base such as tert-butoxide cannot...
-
The COVID-19 pandemic continues to challenge governments and policymakers worldwide. They have rightfully prioritised reducing the spread of the virus through social distancing interventions....
-
What is the relationship between government and the economy, including economic policies, and intervention? Bagheri, M., Guevara, Z., Alikarami, M., Kennedy, C. A., & Doluweera, G. (2018). Green...
-
3.Taxpayer, who is in the highest federal tax bracket in the current year, has a $5,000 gain from a collectible and a $5,000 gain from stock, both held long-term. (a) What is Taxpayer's net capital...

Study smarter with the SolutionInn App


