How might you carry out the following selective transformations? One of the two schemes requires a protection
Question:
How might you carry out the following selective transformations? One of the two schemes requires a protection step. (Recall from Section 19.5 that aldehydes are more reactive than ketones toward nucleophilicaddition)
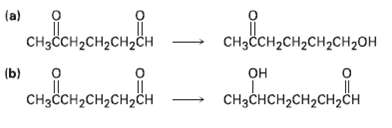
Transcribed Image Text:
"нено (a) одоненоненон CHзCсH-CH2CH2CH2ОH CHзссH2сH2CH2сн (b) "oneнономе онрнененемо CнзсCHн сH2сH2сн CнзCнсH-CH2CH,ҫH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
In general ketones are less reactive than aldehydes for both st...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
How might you carry out the following transformation? More than one step is needed. -CH2 CH2CH2OH
-
How would you carry out the following reactions to introduce deuterium into organicmolecules? (a) CHH2C3CCH2C Hs 2H5 (b) C2H5 CHH2C3CCH2CH3 C=C Hs D (c) CH3CH2CH2C=CH CH3CH2CH2C=CD CD CD2 C=CH (d)
-
1. Evaluate the term "health" and the historical perspective on health promotion. 2. Examine health promotion and illness prevention teaching based on teaching principles, varied teaching learning...
-
How is Netflix an Innovator? What are the consequences to competitors of Netflix's innovations?
-
Write an equation of the ellipse satisfying the given conditions. a. Vertices: (-5, 0), (5, 0); foci: (-2, 0), (2, 0) b. Vertices: (0, -3), (0, 3); length of minor axis: 4 c. Foci: (-3, 0), (3, 0);...
-
What is the Wasson SE Process Model?
-
Delta Aluminums management is considering eliminating product B, which has been showing a loss for several years. The companys annual income statement, in $000s, is as follows: Required a. Restate...
-
Luke Corporation produces a variety of products, each within its own division. Last year, the managers at Luke developed and began marketing a new chewing gum, Bubbs, to sell in vending machines. The...
-
X Co. acquired 75% of Y Co. on January 1, Year 1, when Y Co. had common shares worth $100,000 and retained earnings of $70,000. The acquisition differential was allocated as follows on this date:...
-
When 4-hydroxyhutanal is treated with methanol in the presence of an acid catalyst, 2-mcthoxytctrahydrofuran is formed. Explain. -H C HCH2H2CH C
-
How would you synthesize the following substances from benzaldehyde and any other reagentsneeded? (c) (b) (a) CH- CH2
-
Repeat 6.19.2 for a large disk farm operated by an online backup company. Does upgrading to either RAID 0 or RAID 1 make economic sense given that your income model is based on the availability of...
-
The David Derrick Insurance company wants to sells a certain policy for $1000 to John Graham Jr. They know from historic data that there is a 1 in 100 chance John will file a $20,000 claim, a 1 in...
-
How might the principles of Continuous Improvement, rooted in the ethos of empirical inquiry and relentless introspection, serve as a catalyst for the cultivation of a learning organization paradigm,...
-
Use the information provided in the table to answer the following questions. Assume that there were no changes in any other asset or liability accounts. December 2022 December 2023 Net Income...
-
Robert is planning to open his own residential building design business. What nontax factors should Robert consider as he chooses his business entity? Discuss and explain whether the nontax factors...
-
A stock price is currently trading at $100. Over the next two 6 month periods it will be with up 10% or down 10%. The risk-free interest rate is 8% per annum with continous compunding. What is the...
-
In the calculation of the path integral, we of course have to sum over all possible paths between the initial and final positions. However, it can be useful to consider what contributions to the path...
-
(8%) Problem 6: A student attaches a f= 3.5 kHz oscillator to one end of a metal rail of length L = 25 m. The student turns on the oscillator and uses a piezoelectric gauge at the other end to...
-
Which molecule is nonpolar? (a) SO 3 ; (b) CH 2 Cl 2 ; (c) NH 3 ; (d) FNO.
-
Write structural formulas for both of the possible products from the following Dieckmann condensation, and predict which one would likely predominate. EtOH, heat
-
When a Dieckmann condensation is attempted with diethyl succinate, the product obtained has the molecular formula C12H16O6. What is the structure of this compound?
-
Show how this diketone could be prepared by a condensation reaction:
-
3. Show that : -3t = y 3 sin(t) cos(t) + e is a solution to the differential equation y' + 3 y 10 sin(t)
-
The United States, Austrailia, Canada, and the United Kingdom cluster high on individualism but low on power distance on Hofstede\'s cutlutral graph. What does this mean to someone looking to market...
-
Mini-garden project Week one of the mini-garden project involved Sacha and her fellow pre-schoolers planting sunflower seeds into the outdoor garden bed. Each child in the activity has been assigned...

Study smarter with the SolutionInn App


