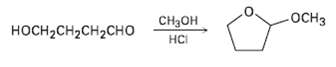
When 4-hydroxyhutanal is treated with methanol in the presence of an acid catalyst, 2-mcthoxytctrahydrofuran is formed. Explain.
Question:
When 4-hydroxyhutanal is treated with methanol in the presence of an acid catalyst, 2-mcthoxytctrahydrofuran is formed. Explain.
Transcribed Image Text:
-ОСHЗ Cнон носHCH2сH2CHо нCі
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
4Hydroxybutanal forms a cyclic hemiacetal when the hydroxyl ox...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
When benzene is treated with I 2 in the presence of C u Cl 2 , iodination of the ring is achieved with modest yields. It is believed that C u Cl 2 interacts with I 2 to generate I + , which is an...
-
On heating 1,2,4-butanetriol in the presence of an acid catalyst, a cyclic ether of molecular formula C4H8O2 was obtained in 81-88% yield. Suggest a reasonable structure for this product.
-
In a sulfuric acid plant, sulfur is burned in the presence of excess oxygen to produce sulfur dioxide, (SO,) which in turn is further reacted in the next step with oxygen in a converter to produce...
-
Analyze how you will use the challenge the process and enable others to act practices to improve the three leadership areas that you selected in module one. ? select one leadership theory or approach...
-
1. What sources of conflict were present in this scenario? 2. On PPT 9-4, The Conflict Process, overt behaviors are depicted as indicators of manifest conflict. What overt behaviors did you identify...
-
Jenna made an investment for 1 year that earned $7.50 simple interest. If the principal had been $25 more and the interest rate 1% less, the interest would have been the same. Find the principal and...
-
What problems with existing Engineering paradigms is it intended to correct?
-
Howe Chemicals Company acquires a delivery truck at a cost of $31,000 on January 1, 2014. The truck is expected to have a salvage value of $4,000 at the end of its 4-year useful life. Compute annual...
-
The makers of a popular cell phone have upgraded its battery capacity from 4500 C to 6300 C. The standard charger can deliver a current of 0.9 A. What is the time taken to charge the phone using the...
-
Mary has been arrested for robbing banks, philosopher _______________ would feel that Mary has lost her person hood and shamed her whole community. Henry gets into a terrible car accident and can no...
-
Aldehydes can be prepared by the Wittig reaction using (methoxymethylene)-triphenylphosphorane as the Wittig reagent and then hydrolyzing the product with acid. For example, (a) How would you prepare...
-
How might you carry out the following selective transformations? One of the two schemes requires a protection step. (Recall from Section 19.5 that aldehydes are more reactive than ketones toward...
-
Let E be an extension field of a finite field F, where F has q elements. Let E be algebraic over F of degree n. Prove that F() has q n elements.
-
A 0.025-kg bullet enters a 2.35-kg watermelon with a speed of 217 m/s and exits the opposite side with a speed of 109 m/s. If the melon was originally at rest, then what speed will it have as the...
-
A girl delivering newspapers covers her route by traveling 8.70 blocks north, 2.18 blocks south, and 9.93 blocks east. Find the angle (a positive value in , between 0 and 360) of her resultant...
-
A remote controlled toy car starts from rest and begins to accelerate in a straight line. The figure below represents "snapshots" of the car's position at equal 0.5 s time intervals. (Assume the...
-
You have invested $100,000 in a badly built house. For $20,000 invested today, you can fix up the house and sell it 1 year from today for $90,000. As an alterna- tive, you can sell the house today...
-
You take a long position in a one-year forward contract on a stock whose price you expect to increase in the future. The current price of the stock is $100 per share. The interest rate is 10%...
-
In this chapter, we only considered path integrals for one-dimensional systems, but in some cases it is straightforward to calculate the path integral for a system in three spatial dimensions. As an...
-
Before the latest financial crisis and recession, when was the largest recession of the past 50 years, and what was the cumulative loss in output over the course of the slowdown?
-
The highest bond-dissociation energy is found in (a) O 2 ; (b) N 2 ; (c) Cl 2 ; (d) I 2 .
-
Write a structural formula for the product from each of the following reactions. (a) (b) (c) (d) (e) (f) NaOEt EtOH NaOEt EtOH NaOEt EtOH NaOEt EtOH (1)NaOEt, EtOH
-
Show all steps in the following syntheses. You may use any other needed reagents but you should begin with the compound given. (a) (b) (c) OEt OEt OEt OEt OEt
-
Provide the starting materials needed to synthesize each compound by acylation of an enolate. (a) (b) (c) CO2Et
-
Bailey Delivery Company, Incorporated, was organized in 2021 in Wisconsin. The following transactions occurred during the year: a. Received cash from investors in exchange for 17,000 shares of stock...
-
Write a program (C++) that prompts the user to input an integer and then outputs both the individual digits of the number and the sum of the digits. Example: Input: 3456 Output: 3 4 5 6 Sum of digits...
-
The soil profile at a site consists of 1.5 m of sand underlain by a 6 m thick clay layer. The water table coincides with the top of the sand layer. The saturated unit weight (Ysat) of the sand and...

Study smarter with the SolutionInn App


