How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
Question:
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen?
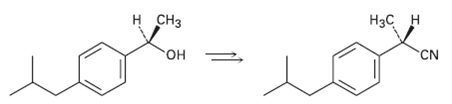
Transcribed Image Text:
н снз Нас н он CN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
Strategy Recall from Chapter 11 that OH is a very poor leaving group in re...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following synthesis (more than one step is required)? What stereo chemical relationship between the ?CO 2 CH 3 group attached to the cyclohexane ring and the ?CHO groups...
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
Jeremy acquired the following ordinary shares in Scarlon plc: He made no further acquisitions during 2021. On 22 December 2020, he sold 10,000 shares in the company for 10 per share. Calculate the...
-
How do functional tactics differ from corporate and business strategies?
-
Solve the following equations, justifying each step by referring to an appropriate property or theorem. (a) 2x + 5 = 8; (b) x2 = 2x; (c) x2 - 1 = 3; (d) (x - 1)(x + 2) = 0:
-
Long, CPA, has been engaged to examine and report on the financial statements of Maylou Corporation. During the review phase of the study of Maylou's system of internal accounting control over...
-
Executives at Meds-R-Us have decided to build a new production facility for the companys best-selling high-blood-pressure drug. The problem they now face is determining the size of the facility (in...
-
Pick an industry in the Resilinc 2018 Annual report. Describe uncertainties in the industry and possible risk mitigation. Search the internet for one or two examples of that industry and how they...
-
Tankers arrive at an oil port with the distribution of interarrival times shown in Table 12. The port has two terminals, A and B. Terminal B is newer and therefore more efficient than terminal A. The...
-
Use the reaction of a Grignard reagent with a carbonyl compound to synthesize the following compound:
-
What product(s) would you expect from dehydration of the following alcohols with POCl3 in pyridine? Indicate the major product in eachcase? (a) (b) (c) CH3CH2CHCHCH3 CH CH3 H (d) (e) CCCH2CH...
-
I dont need to know accounting principles because my business is a sole proprietorship. Comment on this statement.
-
Do you believe you have an internal or external locus of control when it comes to your health? Please explain how this orientation to health may contribute to an individual's decision to get the...
-
Your assignment this week is to do some research on current legislation on policies that support families either local or that are being debated at the federal level. An example is this plan released...
-
Discuss the influence of Stuart Hall in the development of the field of cultural studies.
-
If the president of a country needs some support to arrange governance issues, would you recommend a coach or a mentor ? Which one would be better to use and when to use coach and mentor.
-
When selecting the appropriate supplier, buyers may use which tool to identify the supplier's good and bad qualities? Explain
-
Current assets are listed: a. in the reverse order of expected conversion to cash. b. by importance. c. by longevity. d. by size.
-
Would you expect an ionic solid or a network covalent solid to have the higher melting point?
-
First, complete and balance each of the equations below. Then, choosing among ethanol, hexane, and liquid ammonia, state which (there may be more than one) might be suitable solvents for each of...
-
Dimethylformamide (DMF), HCON(CH3)2, is an example of a polar aprotic solvent, aprotic meaning it has no hydrogen atoms attached to highly electronegative atoms. (a) Draw its dash structural formula,...
-
As noted in Table 3.1, the pKa of acetone, CH3COCH3, is 19.2. In Table 3.1 (a) Draw the bond-line formula of acetone and of any other contributing resonance form. (b) Predict and draw the structure...
-
Write a classification essay in 3 rd person that you can describe clearly and cleverly. Have a clear thesis in the introduction that clearly list their classification categories.
-
China's giant property developer Country Garden has been declared in default on its debt after failing to make a bond repayment by a final deadline of October 18. A panel of global banks and...
-
Why college are important?

Study smarter with the SolutionInn App


