What product(s) would you expect from dehydration of the following alcohols with POCl3 in pyridine? Indicate the
Question:
What product(s) would you expect from dehydration of the following alcohols with POCl3 in pyridine? Indicate the major product in eachcase?
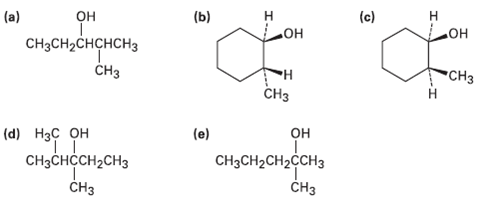
Transcribed Image Text:
(a) он (b) (c) CH3CH2CHCHCH3 Но° но CHз CH3 н СHз он (d) Нзс он (e) CнзCнсCH2CHз CH3CH2CH2CCH3 CHз CHз I.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (22 reviews)
a OH CH3CHCHCHCH32 POCI3 pyridine CH3CH H CH3 H3C H CH3 major H CC OH da POCI3 pyridine CH3 minor Th...View the full answer

Answered By

Benard Ndini Mwendwa
I am a graduate from Kenya. I managed to score one of the highest levels in my BS. I have experience in academic writing since I have been working as a freelancer in most of my time. I am willing to help other students attain better grades in their academic portfolio. Thank you.
4.90+
107+ Reviews
240+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What products would you expect from each of the following reactions? (a) (b) (c) t-BuOK CHBr CH2Zn(Cu) diethyl ether
-
What products would you expect from the reaction of ethylmagnesium bromide (CH3 CH2MgBr) with each of the following reagents? (a) H2O (b) D2O (c) (d) (e) (f) (g) then H O "H Ph" PhPh, then NH4CI, HO...
-
What products would you expect from the reaction of propyllithium (CH3CH2CH2Li) with each of the following reagents? (a) (b) (c) (d) Ethanol (e) H, then H3O , then NH,CI, H20 1-Pentyne, then , then...
-
In April 2016, Vanessa bought 100 shares in Entagon plc at a cost of 5 per share. The company went into liquidation and Vanessa received a first distribution of 40p per share in July 2020. The shares...
-
How does the concept "translate thought into action" bear on the relationship between business strategy and operating strategy? Between long-term and short-term objectives?
-
Although STP marketing often produces successful outcomes, there is no guarantee that these successes will last. What factors can erode the successes produced by STP marketing, forcing a firm to...
-
Which of the following statements about ensemble methods is false? a. In a boosting ensemble, the training data for an individual model depends on the predictions of the previously constructed...
-
M. Anthony, LLP, produces music in a studio in London. The cost of producing one typical song follows: Average Cost per Song: Labor, including musicians and technicians . . . . . . . . . . . . ....
-
Matthew wants to take out a loan to buy a car. He calculates that he can make repayments of $4000 per year. If he can get a five-year loan with an annual interest rate of 4%, what is the maximum...
-
Shek Enterprises is owned by Memphis Shek and has a December 31 fiscal year end. The company prepares adjusting entries on an annual basis. Some additional information follows: 1. A one-year...
-
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
-
What alcohols would give the following products on oxidation? (b) (c) C (a) C
-
The following data are from the Canada Health Survey, and given in Rao and Thomas (1989, p. 107) . They relate smoking status (current smoker, occasional smoker, never smoked) to fitness level for...
-
1) Suppose you have $20,000 in cash to invest. You decide to short sell $10,000 worth of Coca Cola stock and invest the proceeds from your short sale, plus your $20,000, in Intel. At the end of the...
-
What are the CCI Code Pair Edits and PTP modifiers for lab codes 85025, 80053, 87636, 85014?
-
At what velocity (in revolutions per minute) will the peak voltage of a generator be 490 V, if its 480 turn, 8.00 cm diameter coil rotates in a 0.250 T field? rpm +
-
How did Ptolemys atlas contribute to a greater understanding of world geography, and how did it produce confusion?
-
Drinking fountains that actively chill the water they serve cant work without ventilation. They usually have louvers on their sides so that air can flow through them. Why do they need this airflow?
-
Discuss the sampling strategy and technique used to access the appropriate sample. Develop a hypothetical research scenario that would necessitate the use of a 3-Factor Crossover Design. The research...
-
Which should drive action planning more, strengths or weaknesses? That is, is it more important to build on your strengths or to reduce your weaknesses? Explain.
-
Consider the following ions: Na + , K + , Ca 2+ , Mg 2+ , F , Br , O 2 , and S 2 . Which cation and which anion do you expect to combine to form the highest melting compound? Carefully explain your...
-
Formamide (HCONH2) has a pKa of approximately 25. Predict, based on the map of electrostatic potential for formamide shown here, which hydrogen atom(s) has this pKa value. Support your conclusion...
-
Formic acid (HCO2H) has Ka = 1.77 10-4.
-
(a) An acid (HA) has Ka = 10-7. What is its pKa? (b) Another acid (HB) has Ka = 5.What is its pKa? (c) Which is the stronger acid?
-
GDP exceeds NDP by an amount equal to? Explain
-
A not-for-profit hospital reported in 2021 income statement: Revenue 150m Expenses 110m Net Income 40m On their 2021 Statement of Changes in Net Assets they reported: Beginning Net Assets of 110m...
-
Various cost accounting methods record, classify, analyze, and evaluate alternative courses of action used to control costs. The chief information security officer (CISO) uses these results to...

Study smarter with the SolutionInn App


