How would you carry out the following transformations? Tell the reagents you would use in each case.
Question:
How would you carry out the following transformations? Tell the reagents you would use in each case.
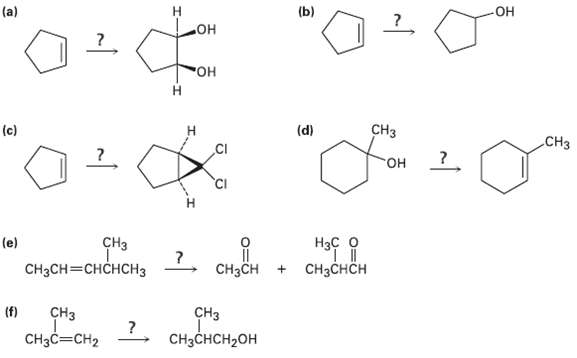
Transcribed Image Text:
(a) (b) Он но° "Он н CHз (c) (d) CHз но- CHз CH3CH=CHCHCH3 (e) Нас о ?. CнзCH + CHзснсH (f) CHз CHз CHзсHCH-оH CH3C=CH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
a b OsO4 NMO H I OH OH OH 1 HgOAc2 H2O 2 NaBH4 Hy...View the full answer

Answered By

Brown Arianne
Detail-oriented professional tutor with a solid 10 years of experience instilling confidence in high school and college students. Dedicated to empowering all students with constructive feedback and practical test-taking strategies. Effective educator and team player whether working in a school, university, or private provider setting. Active listener committed to helping students overcome academic challenges to reach personal goals.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
-
Show that the angular wave number k for a non-relativistic free particle of mass m can be written as in which K is the particle's kineticenergy 27 V2mK k =
-
Calculate each Poisson probability: a. P(X = 6), = 4.0 b. P(X = 10), = 12.0 c. P(X = 4), = 7.0
-
f(x) = ln(l - x) with the base point a = 0. Use your result to find Find the Taylor polynomial of degree n and the Taylor series for the above function. Add up the given series by assuming that the...
-
Explain various types of payment arrangements that impact cash flow.
-
Selected transactions from the journal of June Feldman, investment broker, are presented below. Instructions (a) Post the transactions to T-accounts. (b) Prepare a trial balance at August 31,2017....
-
What are the aims and tools of promotion, and explain why promotion is important for companies. Please explain with definition the promotional tools and briefly describe what are the elements that...
-
You are scheduling production of your popular Rustic Coffee Table. The table requires a top, four legs, 1/8 gallon of stain, 1/16 gallon of glue, 2 short braces between the legs and 2 long braces...
-
Predict the products of the following reactions, showing both regiochemistry and stereochemistry where appropriate: CH (b) (a) 1. 2. Zn, H* M H* CH 1. Hg(OAc)2, H20 2. NABH4 (c) (d) C 1. 2. Hz, "
-
Which reaction would you expect to be faster, addition of HBr to cyclohexene or to 1-methylcyclohexane? Explain.
-
a) Sketch on the same diagram the graphs of y = sin 2 and y = cos for 0 180. b) State the number of roots of the equation sin 2 = cos for which 0 0 180. c) Find the roots of the equation sin 2...
-
Suppose a 20-year Treasury bond with 5% semiannual coupon is currently traded at a yield of 6% (APR). 1) Is the price above or below 100? 2) What is the current price of the bond? 3) If the yield...
-
Alexandra bought a 7-year bond at a 7.50% discount to its par value of $1,000 three years ago. The bond has a 4.70% coupon rate payable semiannually. What was her effective annual rate of return if...
-
Madox Max Ltd pay a dividend of $3.3 per share next year. After that the dividend is forecast to grow at 15% for 1 year. From year three onwards dividends are expected to grow by 6% indefinitely....
-
Consider a coupon bond paying a 8% coupon rate annually, with a face value of HUF10,000, maturing in 6 years. Suppose that the appropriate discount rate is 5%. What is the current value of the bond?
-
What is the difference between enterprise-based and price-based multiples? Give an example of each and explain why they are defined as belonging to each category. 2.Why and how the incentive...
-
Do all trench-dwelling organisms live in hydrothermal vent communities? Explain.
-
Beginning with a country that has a trade deficit, demonstrate graphically what will happen to a countrys potential output with globalization if that countrys costs of production fall. Explain your...
-
Methanol has a normal boiling point of 64.6 C and a heat of vaporization (H vap ) of 35.2 kJ/mol. What is the vapor pressure of methanol at 12.0 C? SORT You are given the normal boiling point of...
-
Predict the products, including their stereochemistry, from the E2 reactions of the following diastereomers of stilbene dibromide with sodium ethoxide in ethanol. Assume that one equivalent of HBr is...
-
Draw the structure of the starting material that would undergo azir-elimination give the -E isomer of the alkene product in the E2 reaction of Eq. 9.40.
-
Draw the structure of the starting material that would undergo azir-elimination give the -E isomer of the alkene product in the E2 reaction of Eq. 9.40.
-
answer the following questions to help me further understand the text. 1. Identify the audience 2. Who needs the analysis of the text that you will communicate in your project? This will be the...
-
I need s fresh set of eyes with a new outlook on this same question that consistently tells me it's already been asked and do I want to unlock that answer. NO! I would like another expert to read my...
-
Do you think Google Corporation is a "monopoly" or is violating antitrust "unfair competition" rules/laws? (Google currently has about 89 percent of the U.S. search engine market, with Bing having...

Study smarter with the SolutionInn App


