How would you prepare the following ethers? (b) CH-CH CH (c) (a) - CH
Question:
How would you prepare the following ethers?
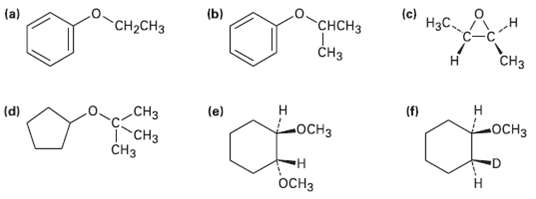
Transcribed Image Text:
(b) НаС "CH-CHз "СНCHЗ (c) (a) -н Снз CHз Н (e) (d) CHз "CHз сHз (f) н -оСнз н ОСНЗ н оСHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
b c OH CHCHCH H3C cc NaH H 1 CH5OH HgOCOCF32 2 NaBH4 CH3 ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you prepare the following ethers using a Williamson synthesis? (a) Methyl propyl ether (b) Anisole (methyl phenyl ether) (c) Benzyl isopropyl ether (d) Ethyl 2, 2-dimethylpropyl ether
-
How would you prepare the following compound using an acetoacetic estersynthesis?
-
How would you prepare the following compound using a Michaelreaction?
-
To pick up the execution results, one can use the executeQuery() and executeUpdate() methods. The former returns an integer, and the latter returns a ResultSet.(True/False)
-
Predict what might have happened to Apple if its top executives had not supported investments in IT.
-
The marketing manager of a large super market chain has the business objective of using shelf space most efficiently. Toward that goal, she would like to use shelf space to predict the sales of a...
-
How should the objective of a variable sampling plan be stated in a two-tail test?
-
Conradt Connectivity Company manufactures various electrical connectors. The companys sales budget for the first six months of the coming year is as follows. All sales are made on credit. Conradt is...
-
Solve each equation for x. (a) x = e-4x=4 (b) In(3x14) = 2 x =
-
You are part of a research team investigating the effects of aerobic cardiovascular and moderate weight resistance training on the BMI (Body Mass Index) of moderately overweight but not obese teens...
-
Predict the products of the following ether cleavagereactions: .CH3 (a) (b) "CH-CH HI CFCO2H CH H20 (d) HI %3DCHH2CH CH CH20-H2CH3 (c) I H20 CH
-
How would you prepare the following compounds from 1-phenylethanol? (a) Methyl 1-phenyl ethyl ether (b) Phenylepoxyethane (c) tert-Butyl 1-phenylethyl ether (d) 1-Phenylethanethiol
-
The following information was extracted from the records of TAC Corporation at the end of the fiscal year after all adjusting entries were completed: Required: 1. Prepare the stockholders equity...
-
Find the compound interest and future value. Round your answers to the nearest cent. Do not round intermediate steps. Principal Rate Compounded $710 5% Daily Time 1 year The future value is $ , and...
-
If you want to double your money in 7 years, what interest rate per year you need to earn? Assume annual compounding.
-
Mary will invest $100 at the end of every month for the next 25 years, starting from next month. What is the future value of her investments at the end of 25 years? Assume an annual interest rate...
-
As a Christian business person, why is it important for you to understand the methods of statistics (ex: average, one dimension picture)? Using one or more scripture passages, explain the value of...
-
You have employment income of $45,000, a business loss of $14,000, capital gains of $20,000, capital losses of $22,000, and subdivision e deductions of $3,000. calculate the income tax , marginal tax...
-
Which of the following items are liabilities of Designer Jewelry Stores? a. Cash. b. Accounts payable. c. Dividends. d. Accounts receivable. e. Supplies. f. Equipment. g. Salaries and wages payable....
-
on 8 For the following set of lengths 130, 170, 160, 160, 150, 190 Third quartile is: et red d out of Select one: O a. 160 a question O b. 145 O c. 175 O d. 180
-
A device is being marketed to your company. The device takes in hot, high-pressure water and generates work by converting it to two outlet streams: steam and low-pressure water. You are asked to...
-
The 1H-NMR spectrum of compound H is as follows, the peaks at highest m/z in the mass spectrum of H appear at 122 and 124 with intensities in a ration of about 1 to 1. Show the structure ofH. 2 H ...
-
Ultraviolet spectroscopy of often used to monitor the amount of a protein in a sample, the amount of protein is correlated with the absorbance at 280nm. Explain which of the following amino acids you...
-
Explain why the ultraviolet spectrum of one of these dienes has its maximum absorption at a longer wavelength than that of the other.
-
Image transcription text 1. A cylindrical specimen of cold-worked copper (see Fig. below) has experienced a ductility of 25%EL. If its cold worked radius is 10 mm, what was its radius before...
-
Question 2 As a project manager, you need to synthesize information about a problem to stakeholders. What should you do first in your synthesizing process? Describe Briefly.
-
Please help me as much as you can!! I will take all your effort to solve this problem and will give you a good rate!!! Please show all the calculations in detail! Also, please do not copied and...

Study smarter with the SolutionInn App


