Predict the products of the following ether cleavagereactions: .CH3 (a) (b) CH-CH HI CFCO2H CH H20
Question:
Predict the products of the following ether cleavagereactions:
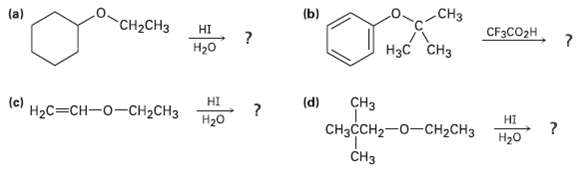
Transcribed Image Text:
.CH3з (a) (b) "CH-CHз HI CFЗCO2H Нас CHз H20 (d) HI Нас%3DCH—о—сH2CHз CHз CняссH2—0-СH2CH3 (c) НI H20 Нао CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (18 reviews)
a b c OCHCH3 OCCH33 HCCHOCHCH3 HI H2O ...View the full answer

Answered By

Mary Njunu
I posses Vast, diversified knowledge and excellent grammar as a result of working in ACADEMIC WRITING for more than 5 years. I deliver work in various disciplines with assurance of quality work. I purpose at meeting the clients’ expectations precisely. Let’s work together for the best and phenomenal grades.
4.90+
928+ Reviews
2551+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions. When more than one product is expected, predict which will be the major product. (a) (b) (c) (d) OH H2SO4 heat H3PO4 heat BrNaocH, CH CH H,SO4 heat OH
-
Predict the products of the following reactions: (a) Excess NH3 + Ph - CH2CH2CH2Br (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) (q) (r) (I) NaN3 (2) LiAIH (3) H30 1-bromopentane CH3...
-
Predict the products of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) OH CI CH NH2 Ph-C-CI+ NH2 0 + (D LiAIH (2) H20 (2) H,0 -. O (I) excess PhMgBr (2) H,o ( CH Mgl (2)...
-
Generally, the JDBC API perform the following three functions a. Connect to database, load JDBC driver, perform the query b. Perform the query, connect to database, load JDBC driver c. Get result...
-
Explain why it would be unethical for Apple to allow its customers to download free music from iTunes.
-
The marketing manager used shelf space for pet food to predict weekly sales. Perform a residual analysis for these data (stored in Pet Food). Evaluate whether the assumptions of regression have been...
-
How does the auditor control the risk of rejecting a fairly stated book value?
-
A firm has experienced a decrease in its current ratio but an increase in its quick ratio during the last three years. What is the likely explanation for these results?
-
The Cantina Restaurant in Reno Nevada has just started a new restaurant in Arizona and has asked you to analyze their operation. Their latest month shows the following results: Number of meals...
-
Sawood Civil Engineering (SCE) is a listed construction company. Its head office is in the capital city, Vaasa, of the country of Nogoland. Nogoland has two neighbouring countries, Alpha and Omega....
-
Give IUPAC names for the followingstructures: (c) CH (b) (a) " SH (f) CH (e) (d) CHCH0- CHH 5. NO2 SCH3 SCH3 (i) (h) CH CH CH3CH2CHCHCHSCHCH3 CH CH (g) CCH H
-
How would you prepare the following ethers? (b) "CH-CH "CH (c) (a) - CH (e) (d) CH "CH H (f) - H
-
Calculate the minimum velocity of flow in ft/s of water at 160F in a 2-in steel tube with a wall thickness of 0.065 in for which the flow is turbulent.
-
If Jason expect a 15 percent portfolio return when objective assessment indicates that its expected return is 8 percent, what cognitive error is Jason demonstrating? Explain
-
What can the slope of the forward curve tell us about storage costs and convenience yields for commodities? Explain
-
Suppose that a (European) call option with strike price $40 costs $13, and a (European) call option with strike price $50 costs $8, and a (European) call option with strike price $60 costs $2. All...
-
What is the relationship between marginal revenue and marginal cost as the firm increases output?
-
If you invested $1,000,000 in a property and received the following cash flows: $100,000 at the end of year one, $95,000 at the end of year two, $105,000 at the end of year three, $80,000 at the end...
-
Presented below is financial information related to the 2025 operations of Delgado Cruises SA. Instructions Prepare the 2025 income statement and comprehensive income statement for Delgado Cruises...
-
What are multinational corporations (MNCs) and what economic roles do they play?
-
A 0.2-m 3 tank containing helium at 15 bar and 22 C will be used to supply 4.5 moles per minute of helium at atmospheric pressure using a controlled adiabatic throttling valve. a. If the tank is...
-
Compounds A and B are isomers with the formula C3H6O. A has a peak at 1730 cm ?1 in its IR spectrum and B has a peak at 1715cm ?1 , the mass spectra of A and B are as follows, show the structures of...
-
Compounds C and D are isomers with the formula C9H12, in addition to other absorption peaks, both compounds show a peak near 7.25 ? (area 5) in their 1H-NMR spectra. Their mass spectra are as follow,...
-
Compounds E and F are isomers with the formula C6H8 both react with H2 in the presence of Pt to give G (C6H12), G shows a single peak in its 13C-NMR spectrum. E has no absorption maximum above 200nm...
-
A couple obtained a $20,000 mortgage loan at an interest rate of 10.5% compounded monthly. (Original principal equals to PV of all payments discounted at the interest rate on the loan contract) (1)...
-
What strategies and tactics are employed to manage strategic risks and uncertainties, including geopolitical instability, supply chain disruptions, and emerging competitive threats, while preserving...
-
How do strategic planners integrate ethical considerations and sustainability imperatives into strategic planning processes, balancing short-term financial objectives with long-term societal and...

Study smarter with the SolutionInn App


