Identify the acids and bases in the followingreactions: CH2 (a) CH3 + Ht TICIA () + TICI,
Question:
Identify the acids and bases in the followingreactions:
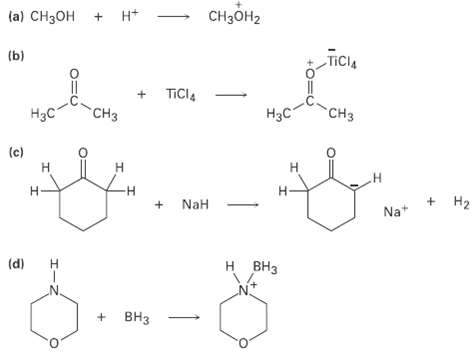
Transcribed Image Text:
CHзон2 (a) CH3ОН + Ht TICIA (ы) + TICI, CHз Нас "CHз Нас (c) Н Н Н + NaH +. Na+ Н2 (d) н внз Н + ВНз т т т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
Reactions a and c are reactions between BrnstedLowry acids a...View the full answer

Answered By

John Aketch
I have a 10 years tutoring experience and I have helped thousands of students to accomplish their educational endeavors globally. What interests me most is when I see my students being succeeding in their classwork. I am confident that I will bring a great change to thins organization if granted the opportunity. Thanks
5.00+
8+ Reviews
18+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify conjugate acids and bases in the following pairs of substances: (a) (HOCH), CNH (HOCH CNH (b) HOCH, CH, N N CH, CH, SO HOCH, CHN N CH, CH, SOs (c) O SCH, CH, N, N+CH, CH, SOg O SCH, CH,N N...
-
Identify the conjugate acids and bases in the following pairs of substances: (a) (CH 3 ) 3 NH + / (CH 3 ) 3 N (b) + H 3 NCH 2 COOH/ + H 3 NCH 2 COO - (c) + H 3 NCH 2 COO /H 2 NCH 2 COO - (d) OOCCH 2...
-
Identify ways in which French and Raven's Five Bases of Social Power are used in your organization (Walmart Supercenter). Address the bases of power individually, not just in general. Evaluate...
-
How does the trade-off between decision management and decision control affect the form that an absorption cost system takes within a particular firm?
-
Which of the following individuals qualify for the earned income credit for 2016? a. Thomas is single, is 21 years of age, and has no qualifying children. His income consists of $9,000 in wages. b....
-
The following data were obtained when a Ca 2+ ion-selective electrode was immersed in standard solutions whose ionic strength was constant at 2.0 M. (a) Prepare a calibration curve and find the...
-
When creating association rules, why is it common to only consider rules with a support measure above a minimum threshold? a. Rules with too low of a support are unlikely to be true. b. Rules with...
-
Your firm is considering a new investment proposal and would like to calculate its weighted average cost of capital. To help in this, compute the cost of capital for the firm for the following: a. A...
-
Allen Corp. enters into a contract with a customer to build an apartment building for $1,031,300. The customer hopes to rent apartments at the beginning of the school year and provides a performance...
-
Professor Siegel argues that investing in stocks for retirement may be less risky than investing in bonds. Would you recommend this approach to an individual in his or her early 60s?
-
Assume that you have two unlabeled bottles, one of the following which contains phenol (pKa = 9.9) and one of which contains acetic acid (pKa = 4.76). In light of your answer to Problem 2.51, suggest...
-
Which of the following pairs represent resonance structures? (a) CH3C=N-O: and CH3C=N-o: (b) :0: :C C0: and (d) (c) : and :CH2-N CH2=N NH3 and NH2
-
You have just learned that your companys board of directors is considering a change in the companys strategic direction that would result in a partial shutdown of the companys local manufacturing...
-
Suppose you have a 4.50% coupon bond with a ytm of 2.0 percent and a term-to-maturity of 3 years.The bond pays its coupon ANNUALLY (once per year) and has a face value of $1,000. What is this bond's...
-
Identify a narrow policy issue of your choice in the Education sector of South Africa. 1. This policy should be attracting negative publicity from international or regional or national stakeholders...
-
A pint of beer costs GBP 4.54 in London, and the spot exchange rate is GBP 0.75 per USD. How many US dollars will it cost you to buy a pint of beer in London? Please round your answer to the nearest...
-
1. Consider the following 6 workers. Their names are in column (1) and their respective utility functions are in column (2). a. The table. I've filled out Adam's row for you. Fill out the rest of the...
-
Consider the following (X, Y) data: (53, 37), (34, 26), (10, 29), (63, 55), (28, 36). Find the regression equation
-
Based on her investigation, Al-Khalili would most likely recommend: A. active currency management. B. a hedging ratio closer to 100%. C. a narrow discretionary band for currency exposures. Kalila...
-
The value of a share of common stock depends on the cash flows it is expected to provide, and those flows consist of the dividends the investor receives each year while holding the stock and the...
-
In this chapter, from the third law of thermodynamics, it has been shown that the entropy of all substances approaches a common value at 0 K (which for convenience we have taken to be zero). This...
-
Explain whether this reaction would follow the SN1 or the SN2 mechanism and then explain which reaction is faster: CI a) CHCH + OH HO CHOH CH3 b) CHC-Br + CHCO CH, O-SOCH3 or CHCH CH CO,H or + OH CH3...
-
(a) Show all the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of electrons in each step of the mechanism. (b) Show a free energy versus reaction...
-
Explain which of these reaction proceeds at a faster rate: CH3 a) CHC-Cl + CHCOH CH Br b) CHCHCHCH CH3O Br c) CHCH,CH,CH, + CHO CH CO,H CHOH CHOH CH3 O or CHC CI+ CHCO CH3 Br CH3COH or CHCHCHCH +...
-
003 The minimal polynomial associated with the matrix 102 01 1 is
-
Question 1: How do strategy and tactical action relate to each other in an organization? Question 2: How can you improve the implementation of strategy in this business organization?...
-
Larkin Corporation sells products for $125 each that have variable costs of $50 per unit. Larkin's annual fixed costs are $430,000. (10 points Total) a. What is Larkin's contribution margin per unit?...

Study smarter with the SolutionInn App


