Identify the chirality centers in the following molecules (yellow-green = Cl, pale yellow =F): (b) (a) Threose
Question:
Identify the chirality centers in the following molecules (yellow-green = Cl, pale yellow =F):
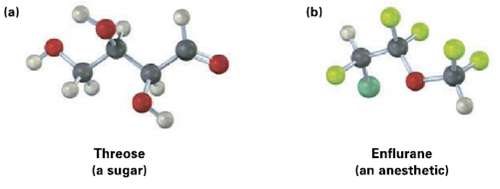
Transcribed Image Text:
(b) (a) Threose (a sugar) Enflurane (an anesthetic)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a H H H ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the chirality centers in the following molecules. Build molecular models if you needhelp. (b) (c) CH30. CH2CH2CH3 C (a) - Coniine (poison hemlock) N-CH3 Menthol (flavoring agent)...
-
Assign R or S stereochemistry to the chirality centers in the following Newmanprojections: CI (a) (b) C "CH . "
-
Assign R or S configurations to the chirality centers in the followingmolecules: CI H la) (c) (b) H
-
Outline suggestions to make observation a useful, reliable requirements elicitation technique.
-
Discuss why sound travels faster in moist air. (Note that at the same temperature, water vapor molecules have the same average kinetic energy as the heavier nitrogen and oxygen molecules in the air.)...
-
A simply supported beam AB supports a trapezoidally distributed load (see figure). The intensity of the load varies linearly from 50 kN/m at support A to 25 kN/m at support B. Calculate the shear...
-
A system, consisting of \(2 \mathrm{~mol}\) of \(\mathrm{N}_{2}, 5 \mathrm{~mol}\) of \(\mathrm{H}_{2}\) and \(2 \mathrm{~mol}\) of \(\mathrm{NH}_{3}\) initially, is undergoing the following...
-
Hovak Company has credit sales of $ 4.5 million for year 2013. At December 31, 2013, the companys Allowance for Doubtful Accounts has an unadjusted debit balance of $ 3,400. Hovak prepares a schedule...
-
A market for private math tutors in Madison organized by the Students' Union. What is the wage rate that math tutors earn and how many are hired? If the Students' Union sets the minimum wage for...
-
The National Gang Crime Research Center (NGCRC) has developed a six-level gang classification system for both adults and juveniles. The six categories are shown in the accompanying table. The...
-
Alanine, an amino add found in proteins, is chiral. Draw the two enantiorners of ala- nine using the standard convention of solid, wedged, and dashedlines. NH2 Alanine CHO2H
-
Is cocaine (Worked Example 9.2) dextrorotatory or levorotatory?
-
After Island Farms increased the container size for its premium ice cream from 1.65 L to 2.2 L, the retail price increased from $5.49 to $7.98. What was the percent change in the unit price?
-
How would you perform a test of the predictions of the capital assest pricing model given historical data on asset and market portfolio returns?
-
Critique the accuracy of all estimates and explain some of the factors that determine the firms' equity, debt and therefore weighted average cost of capital. Keuring Keycrop Risk Free Return 3.87%...
-
A small oil company, Hastelloy, uses micro - moving Coriolis axle gauges on its construction site. Emerson wants to replace it with F series axle meters. This replacement process will apply to the...
-
1. One assumption underlying Modigliani and Miller's theories of capital structure is that there is no difference between 'home - made' gearing and corporate gearing. Explain this assumption and...
-
Discuss the goal of financial management around the world from finance marketing and professional perspective?
-
From a distance of 4.0 m, a bystander listens to a jackhammer breaking concrete. How far would he need to move from the jackhammer so that its perceived loudness decreases by a factor of 8?
-
What are some of the features of the Unified Process (UP)?
-
Methanol (CH 3 OH) can be synthesized by the reaction: What volume (in liters) of hydrogen gas, at a temperature of 355 K and a pressure of 738 mmHg, is needed to synthesize 35.7 g of methanol? CO(g)...
-
Predict the product when each of the following compounds reacts with one equivalent of lithium dimethylcuprate, followed by protonolysis. Explain. CH,(CH,),0-C(CH,),C-CI
-
Outline two methods for the preparation of 5-methylhexanoic acid from 1-bromo-4-methylpentane.
-
One interesting process for making nylon-6,6 demonstrates the potential of using biomass as an industrial starting material. The raw material for this process, outlined in the following reaction, is...
-
ABC Hospital is one of two hospitals in the community. The Hospital offers all major specialty services (e.g. cardiology, neurology, pulmonology, etc.) The Hospital is approached by a cardiologist...
-
You obtained job-related information about the HR assistant position from O*Net. Answer the following sections based on that information. QUESTIONS: The following is the list of questions included in...
-
On January 8, the end of the first weekly pay period of the year, Regis Company's employees earned $27,760 of office salaries and $70,840 of sales salaries. Withholdings from the employees' salaries...

Study smarter with the SolutionInn App


