Identify the indicated faces in the following molecules as Re orSi: (b) (a) H-c-CH3 CO2 02C-CH
Question:
Identify the indicated faces in the following molecules as Re orSi:
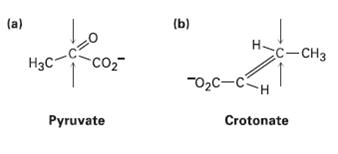
Transcribed Image Text:
(b) (a) H-c-CH3 CO2 Нас "02C-CH Crotonate Pyruvate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
a Re face R H...View the full answer

Answered By

HILLARY KIYAYI
I am a multi-skilled, reliable & talented Market analysis & Research Writer with a proven ability to produce Scholarly Papers, Reports, Research and Article Writing and much more. My ultimate quality is my English writing/verbal skill. That skill has proven to be the most valuable asset for project writing, Academic & Research writing, Proofreading, HR Management Writing, business, sales, and a variety of other opportunities.
4.80+
24+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the indicated hydrogen?s in the following molecules as pro-R or pro-S: (a) (b) .co2 H3N Alanine (S)-Glyceraldehyde
-
Identify the indicated protons in the following molecules as unrelated, homotopic, enantiotopic, ordiastereotopic: (a) (b) Cysteine
-
Identify the indicated hydrogens in the following molecules as pro-R or pro-S: (c) (a) (b) CH3S, .co2- - HS N H3N H Cysteine Malic acid Methionine
-
As a brand manager, for a product of your choice, develop a strategy for launching the product in a manner most likely to capture the attention of the product's primary target market. Write a...
-
Discuss how the conservation of momentum is a consequence of Newton's third law?
-
The following data were extracted from the accounting records of Sacajawea Mercantile Co. for the year ended June 30, 20Y4: a. Journalize the June 30, 20Y4, adjusting entry for estimated sales...
-
What are some potential disadvantages of using a packaged application to help automate the employee goal-setting process?
-
Humble Company has provided the following budget information for the first quarter of 2016: Total sales ....................$ 208,000 Budgeted purchases of direct materials ....... 40,150 Budgeted...
-
discuss the role of ethical leadership in building resilience and guiding organizations through times of crisis or uncertainty, by prioritizing ethical considerations and maintaining a steadfast...
-
Elm is a public company with a 12/31 fiscal year-end. Elm reported a 12/31/2020 debit balance in AOCI of $53,000. Elm Inc. reported net income for the fiscal year 2021 of $140,000. In addition, the...
-
Answer Problem 9.61 for the epoxidation of trans-4-octene. CH3CH2CH2CH=CHCH2CH2CH3 CH3CH2CH2CH-CHCH2CH2CH3 RCO3H 4-Octene 4,5-tane
-
Draw all possible stereo isomers of 1, 2-cyclobutanedicarboxylic acid, and indicate the interrelationships. Which, if any, are optically active? Do the same for 1, 3-cyclohutanedicarhoxylic acid.
-
(a) Discuss the main structural features of the electrical double layer. (b) Distinguish between the electrical double layer and the Nernst diffusion layer.
-
This problem presents a variation of the rancher-farmer example that we discussed in class. Suppose that a rancher faces a choice of how many cattle to put on his property. If he puts more than 21,...
-
Sometimes the governments of free market countries try to control prices to support an industry rather than the functions of supply and demand. For example, the US government supports various...
-
At the end of the current year, Singleton Inc. reported the following information: Sales $412,000 Operating income 163,000 Average total assets 475,000 Average stockholders' equity 365,000. Compute...
-
Watchdog to monitor soaring cost of flights as airlines keep supply low By Mark Saunokonoko, Senior Journalist, Dec 6, 2022 Australia's consumer watchdog has warnedairlinesthat the price of their...
-
What is a transaction in SQL? How do you ensure ACID properties in database transactions?
-
How much of Dodges board-designated assets should be included in net assets without donor restrictions? a. $0 b. $40,000 c. $700,000 d. $740,000 Under its established rate structure, Dodge Hospital...
-
Sandcastles, Inc.s management has recently been looking at a proposal to purchase a new brick molding machine. With the new machine, the company would not have to buy bricks. The estimated useful...
-
An acid solution is 0.100 M in HCl and 0.200 M in H 2 SO 4 . What volume of a 0.150 M KOH solution would completely neutralize all the acid in 500.0 mL of this solution?
-
Rank the following compounds in order of increasing reactivity toward nitration with HNO3 and explain your choices: thiophene, benzene, 3-methylthiophene, and Z-methvlfuran.
-
Think of the compounds in the following sets as enols. Then draw the carbonyl isomers of the following compounds. Which compound within each set contains the greatest percentage of carbonyl isomer?...
-
Rank the compounds within each of the following sets in order of increasing basicity, and explain your reasoning. (a) Pyridine, 3 - nitropyridine, 3 - chlorogyridine (b) imidazole and thiazole
-
A system consisting of a small 1.20-kg object attached to a Ax(cm) light spring oscillates on a smooth, horizontal surface. A 5.00- graph of the position x of the object as a function of time is 4.00...
-
Transform following Relational model into ER Model. id Author writes authorid bookid Book bookid title edition libid memberld name dob AuthorContact id contact publishes Library id bid date id...
-
State suitable case for each model Hierarchical Model Network data model Relational model Object-oriented data model

Study smarter with the SolutionInn App


