In addition to the reaction shown on p. 353, Diphenhydramine can also be prepared by heating bromo
Question:
In addition to the reaction shown on p. 353, Diphenhydramine can also be prepared by heating bromo diphenyl methane and 2-(dimethyl lamino)-1-ethanol in a polar solvent. Show a mechanism for thisreaction:
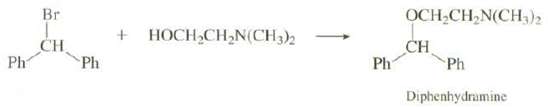
Transcribed Image Text:
Br OCH,CH,N(CH,)2 HOCH CH,N(CH3)2 CH Ph CH Ph Ph Ph Diphenhydramine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
Ph Br CH Ph SN1 ...View the full answer

Answered By

Issa Shikuku
I have vast experience of four years in academic and content writing with quality understanding of APA, MLA, Harvard and Chicago formats. I am a dedicated tutor willing to hep prepare outlines, drafts or find sources in every way possible. I strive to make sure my clients follow assignment instructions and meet the rubric criteria by undertaking extensive research to develop perfect drafts and outlines. I do this by ensuring that i am always punctual and deliver quality work.
5.00+
6+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Diphenhydramine can also be synthesized by heating bromo diphenyl methane with the amino alcohol shown here. Offer a reason why the oxygen, rather than the nitrogen, of this compound acts as the...
-
In addition to the FASB's statement on accounting for research and development activities, the EITF has addressed three implementation issues. List and briefly summarize each of these issues.
-
In addition to the proton marked Ha in m-nitrostyrene in Figure 13.18, there are two other vinylic protons. Assuming that the coupling constant between the two geminal protons in ArCHoeCH2 is 2 Hz...
-
What is the type of the expressions computed on these two lines? 4 > 5 print (4>5)
-
For each of the costs listed below, indicate whether it is: (a) A product or period cost (b) A variable or fixed cost (c) A manufacturing or non-manufacturing cost. 1. Advertising costs of Nike. 2....
-
Tax Return Problem 9: Partnership Instructions: Please complete AAA Fast Plumbing's 2019 tax return based upon the facts presented below. If required information is missing, use reasonable...
-
What is a retention and why is it used in managing project work?
-
Mary Givens and Peggy Moser are partners engaged in operating The G&M Doll Shop, which has employed the following persons since the beginning of the year: T. Binn (general office worker) . . . . . ....
-
Write method call colorMixer, which has two parameters of type char (representing two primary color). The method will return a string, which is the mixed color of the two primary colors given. The...
-
Childrens Best Hope (CBH) provides day care ser-vices to low income families. CBH bills the state for its services under a service contract. Billings for the first four months of 2013 are anticipated...
-
Suggest a mechanism for thisreaction: CH3 CH, CH3 CH3 N. CHCI CI
-
Another Diphenhydramine synthesis is shown in the following equation: (a) Show a mechanism for the first step in this synthesis. (b) Explain which mechanism is occurring in the secondstep. OCH CH...
-
Show that f(x) = x 2 + 8x - 2 is irreducible over Q. Is f(x) irreducible over R? Over C?
-
Suppose that two countries, initially in autarchy, decide to create a single market for only one product. Demand for this product is given by Q(p) = 21 - p in country 1, and by Q(p) = 42-2p in...
-
Consider a representative individual who lives for TWO periods. The individual is endowed with one unit of labour in each period of his/her life. The individual consumes in both periods and his/her...
-
3.31 The output elasticity of demand for input x; is defined as y (w, y) = (ax(w, y)/ay) (y/x;(w, y)). (a) Show that iy (w, y) = (y)eiy (w, 1) when the production function is homothetic. (b) Show...
-
Henri and Lila are having second thoughts regarding their choice of a home mortgage. They thought they should just get a 15-year mortgage, but now they are not so sure. The restaurant is doing...
-
Show (using $ figures) how your business will be able to service its dues or offer suitable ROI. #per Month 4 Project A S Project B 6 Project C 7 Project D $ Cost per 9 Month 10 Project A 11 Project...
-
Identify the four bases of jurisdiction under the UCCJA and indicate how the UCCJEA and PKPA refine them.
-
Show that the peak of the black body spectrum as a function of ? is given by eq. (22.14) kg T Wmax = 2.82
-
Solve each system by graphing. 2.x +3y = 6 4.x + 6.y = 12 -4 -3 in # 3 10 + || + 15 fet en - HAT
-
Draw an energy diagram for the three molecular orbitals of the cyclopropenyl system (C3H3). How are these three molecular orbitals occupied in the cyclopropenyl anion, cation, and radical? Which of...
-
Cyclopropanone is highly reactive because of its large amount of angle strain, but methylcyclopropenone, although even more strained than Cyclopropanone, is nevertheless quite stable and can even be...
-
Cycloheptatrienone is stable, but cyclopentadienone is so reactive that it can?t be isolated. Explain, taking the polarity of the carbonyl group into account. Cycloheptatrienone Cyclopentadienone
-
Students must useinformation literacy in their writing. Theprojectshould be at least #2000 words of content. Word document only! The students will be required to meet with a top level manager,...
-
Your agency has been directed by community leaders to develop and implement a community paramedic program targeting injury prevention among the elderly. You are now tasked with providing a briefing...
-
Write up an assessment record for one of the children/young people in a learning environment. The record must be set out clearly showing: - a) Your assessment of the learner's development b) How the...

Study smarter with the SolutionInn App


