In the iodoform reaction, a triiodomethyl ketone reacts with aqueous NaOH to yield a carboxylate ion and
Question:
In the iodoform reaction, a triiodomethyl ketone reacts with aqueous NaOH to yield a carboxylate ion and iodoform (triiodomethane). Propose a mechanism for thisreaction.
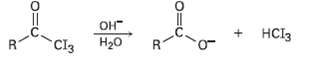
Transcribed Image Text:
OH H20 Cl3 HCI3 R.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
This is a nucleophilic acyl substitution reaction who...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Propose a mechanism that shows why p-chlorotoluene reacts with sodium hydroxide at 350 C to give a mixture of p-cresol and m-cresol.
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Consider the following function. def f(x): a = [] while x > 0: a.append(x) f(x-1) A new object of type list is created for each recursive invocation of f. True or False
-
Explain how blogs and social media networks can be used by businesses and report writers.
-
Find the best fitting line for the logarithm of population 1 as a function of time and compute r2. Is this a better fit? Consider the following data on the growth of two bacterial populations. Year...
-
Explain what kinds of problems the Copycat architecture can solve.
-
Hinshaw Company purchased a new machine on October 1, 2014, at a cost of $90,000. The company estimated that the machine has a salvage value of $8,000. The machine is expected to be used for 70,000...
-
Quantitative Problem: Bellinger Industries is considering two projects for inclusion in its capital budget, and you have been asked to do the analysis. Both projects' after-tax cash flows are shown...
-
Adamantine Architectonics. Adamantine Architectonics consists of a U.S. parent and wholly owned subsidiaries in Malaysia (A-Malaysia) and Mexico (A-Mexico). Selected portions of their...
-
Epoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with epichiorohydrin forms a ?prepolymer,? which is then ?cured? by treatment with a triamine such as H 2...
-
Draw structures for the enol tautomers of the following compounds: (a) Cyclopentanone (b) Methyl thioacetate (c) Ethyl acetate (d) Propanal (e) Acetic acid (f) Phenyl acetone
-
Ling Corporation uses the equity method to account for its ownership of 35% of the common stock of Gorman Packing. During 2020, Gorman reported a net income of $80,000 and declares and pays cash...
-
Identify, analyze, and explain strategies for doing business globally and in relation to international law. Discuss the different ways that exist for a business to operate internationally.
-
10. First, lets look at the working decision of an individual. max{(1-1)+(1-1)w 1; +T_) subject to 1, 0 where is the tax rate and T is the transfer received from the other person. With the same...
-
A business firm is considering hiring 10 additional people to operate an additional production line. The firm calculates that the extra cost of hiring the people is $250,000 and the extra benefit...
-
Intel and AMD, the primary producers of computer central processing units (CPUs), compete with one another in the mid range chip category (among other categories). Assume that global demand for...
-
The expected extra benefit of an additional gallon of gasoline to Norton and Egor is shown below. Gallons of Gas Extra benefit in dollars Norton Ed 1st 2.75 2.00 2nd 1.65 1.40 3rd 1.35 1.00 4th 1.10...
-
Which of these does not belong in your mineral collection? (a) table salt (b) diamond (c) quartz (d) coal
-
A liquid flows upward through a valve situated in a vertical pipe. Calculate the differential pressure (kPa) between points A and B. The mean velocity of the flow is 4.1 m/s. The specific gravity of...
-
Explain the difference between hybrid atomic orbitals in valence bond theory and LCAO molecular orbitals in MO theory.
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
Within each set, which compound should show NMR absorptions with the greater chemical shifts? Explain your choices. (1) (2)
-
Overall, property taxes constitute a small piece of the macroeconomic pie in industrialized countries, but they are an important source of revenue for local governments. To what extent do local...
-
Two charges, q=43.5 nC and qB=-25.5 nC are placed as shown in the picture. y [m] + 9 B P 9A 1 0-1- x [m] What is the magnitude of the net electric field at point P? Provide your answer in N/C, use at...
-
Simplify. 9u6x4 4 3 3wx +9x

Study smarter with the SolutionInn App


