Into how many peaks would you expect the 1H NMR signals of the indicated protons to be
Question:
Into how many peaks would you expect the 1H NMR signals of the indicated protons to be split? (Yellow-green =Cl.)
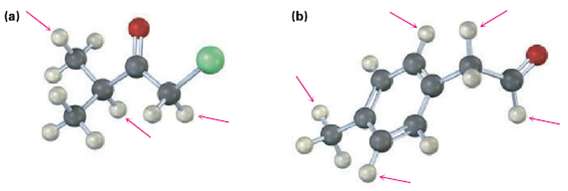
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (19 reviews)
a HC H3C H 00 CHCl 1 dou...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many peaks would you expect to see in the 1H-decoupled 13C NMR spectrum of a. 2-methyl-2-propanol? b. 4-methylcyclohexanone? c. 2-methyl-1-propanol? d. cis-1,3-dimethylcyclopentane?
-
How many peaks would you expect in the 1H NMR spectrum of 1, 4-dimethyl-benzene para-xy1ene or p-xylene)? What ratio of peak areas would you expect on integration of the spectrum? Refer to Table 13.3...
-
How many 1H NMR signals would you expect from each of the following compounds? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) OH CI
-
In a recombinant protein expression process the host microorganism, Pichia pastoris needs to be separated from the culture supernatant after production. A pilot scale bioreactor with a working volume...
-
"The only way to succeed in a market with homogeneous products is to produce more efficiently than most firms." Comment. Does this imply that efficiency is less important in oligopoly and monopoly...
-
The following data present the temperatures at which certain vapor pressures are achieved for dichloromethane (CH2Cl2) and methyl iodide (CH3I): (a)Which of the two substances is expected to have the...
-
What are the most controversial terms of the term sheet? Why are these terms considered to be controversial?
-
Variable Costing Income Statement; Explanation of Difference in Net Operating Income Refer to the data in Exercise 71 for Ida Sidha Karya Company. The absorption costing income statement prepared by...
-
Using economic analysis, fully explain how both private efficiency and social efficiency operate in a tort system of legal rights and remedies. In your answer, fully explain how private costs, social...
-
For the grid shown in Figure P5-48, determine the nodal displacements and the local element forces. Let E = 30 Ã 106 psi, G = 12 Ã 106 psi, I = 200 in.4, and J = 100 in.4 for both...
-
How could you use 1H NMR to determine the regiochemistry of electrophilic addition to alkenes? For example, does addition of HCI to 1-methylcyclohexene yield 1-chloro-1-methylcyclohexane or...
-
How many absorptions would you expect the following compound to have in its 1H and 13C NMRspectra?
-
Clarissa sells her cupcakes for $2.50 each. Clarissa's variable costs per cupcake equal $0.50 and her monthly fixed costs are $3,000. Required: a. What is Clarissa's contribution margin per cupcake?...
-
A payment settlement entity (PSE) must submit Form 1099-K - Transactions with Payment Cards and Third Party Networks for payments made in the settlement of reportable payment transactions for each...
-
f(x) = 3x-5x, find f'(-2)
-
Dave and his spouse, Stefanie, (both over 65) file a joint return for 2023. They have an adjusted gross income (AGI) of $28,000 (and no tax-exempt income or foreign income) and receive benefits from...
-
A 125.0 kg fullback is running at 6.56 m/s toward the goal line. A linebacker with a mass of 80.5 kg is the last one in a position to stop the touchdown. How fast would the linebacker have to be...
-
A gamma ray with an energy of 3.40 10 -14 joules strikes a photographic plate. We know that Planck's constant is 6.63 10 -34 joule seconds. What is the frequency of the photon?
-
On a rifle that has a telescopic sight, the telescope is usually not parallel to the barrel of the rifle. The angle the telescope makes with the barrel has to be adjusted for the distance to the...
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
A solution contains 4.08 g of chloroform (CHCl 3 ) and 9.29 g of acetone (CH 3 COCH 3 ). The vapor pressures at 35 C of pure chloroform and pure acetone are 295 torr and 332 torr, respectively....
-
The NMR spectrum of cinnamaldehyde follows. (a) Determine the chemical shifts of Ha, Hb and Hc. The absorption of one of these protons is difficult to see; look carefully at the integrals. (b)...
-
Consider the proton NMR spectrum of the following ketone. (a) Predict the approximate chemical shift of each type of proton. (b) Predict the number of NMR peaks for each type of proton. (c) Raw a...
-
Use the imaginary replacement technique to show that protons Hc and Hd in cyclobutanol are diastereotopic.
-
6. Decarburization is the decrease of the carbon content of (the surface of) a steel due to interactions with the environment at elevated temperatures. This will result in a layer on the surface with...
-
Discussion Topic Activity Time: 3 Hours Directions: Think of the company and the industry you currently work for. (If you aren't currently working, consider your previous employer.) Discuss how your...
-
Research your industry and find common ratios significant to your industry. What are they and what are they telling you about your industry? Be specific as to the ratios relevant to your industry and...

Study smarter with the SolutionInn App


