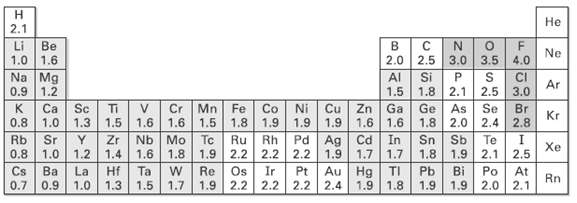
Use the electro-negatively values shown in Figure to rank the following bonds from least polar to most
Question:
Use the electro-negatively values shown in Figure to rank the following bonds from least polar to most molar: H3C ? Li, H3C ? K, H3C ? F, H3C ? MgBr, H3C ? OH.
When companies need to raise money, issuing bonds is one way to do it. A bond functions as a loan between an investor and a corporation. The investor agrees to give the corporation a specific amount of money for a specific period of time in exchange...
Transcribed Image Text:
Не 2.1 Li Be 1.0 1.6 Na Mg 0.9 1.2 Ca 0.8 Ne 4.0 2.0 2.5 3.0 3.5 Al 1.5 Si 1.8 2.1 2.5 Ge As 1.8 2.0 2.4 CI Ar 3.0 Mn Fe V Cr 1.6 1.5 1.8 Sc Ti 1.3 Ga Cu Zn 1.6 Br Se Ni Co 1.9 Kr 2.8 1.0 Rb 1.5 1.9 Rh Pd 2.2 2.2 1.9 1.6 1.6 In Ag Cd 1.7 Zr Nb Mo| Tc Ru 1.6 Sr 0.8 Sb Sn 1.9 2.1 Xe Te 2.5 1.0 1.2 1.4 La Hf 1.0 1.3 1.5 1.8 2.2 Re Os 1.9 2.2 1.7 1.9 1.9 1.7 1.8 Bi Pb Po 1.9 1.9 2.0 Au Hg Ba Cs 0.7 TI At Rn 2.1 Ta Ir Pt 2.2 2.2 2.4 1.9 1.8 0.9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 91% (12 reviews)
Strategy Use Figure to find the electronegativities of each element Calcula...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Use the electro negatively table (Figure) to predict which bond in each of the following sets is more polar, and indicate the direction of bond polarity for each compound. (a) H3C ? C1 OR C1 ? C1 (b)...
-
Modify the diagram shown in Figure to include feed forward command compensation with a constant compensator gain Kf. Determine whether such compensation can eliminate steady-state error for step and...
-
An engineer has proposed the circuit shown in figure to filter out high-frequency noise. Determine the values of the capacitor and resistor to achieve a 3-dB voltage drop at 23.16kHz. ww R
-
What have researchers found about the use of job costing to record the cost of handproducing a bound book?
-
Harriet, an organic farmer, has owned depreciable farm equipment for several years. Is the equipment a capital asset? Why or why not?
-
The variable of interest is the total semester hours of college credit for each candidate. Recall that the mean and median for the data set were 141.31 and 140 hours, respectively, and the standard...
-
In your audit of the Learned Corporation you request a management "rep" letter. You are particularly interested in management's representations on 1. Irregularities involving management or employees....
-
Video Company, Inc., produces and markets two popular video games, High Ranger and Star Bounder. The closing account balances on the companys balance sheet for last year are as follows: Cash,...
-
There are two Excel sheets in the workbook: budgeted and actual. The numbers on the actual sheet come from an external file sent over from another department Monthly. How can the monthly numbers be...
-
The managers of Roosevelt's (a local yet upscale bar) are considering charging an admission fee on Thursday nights. They contemplate how to charge. Should they: Option 1. Use just a beverage charge...
-
Use the + / convention to show the direction of expected polarity for each of the following bonds indicated. (a) H3C C1 (b) H3C NH2 (c) H2N H (d) H3C SH (e) H3C MgBr (f) H3C F
-
Look at the following electrostatic potential map of chloromethane, and tell the direction of polarization of the C ? C1 bonds: CI C-H Chloromethane H.
-
Distinguish between AIS and MIS.
-
Solve the following logarithmic equation for x , if it exists: log 2 ( x ) + log 2 ( x - 1 ) = 1 . Find it to two decimal points
-
Find the value of x which satisfies the following equation. log 2 ( x 1 ) + log 2 ( x + 5 ) = 4
-
Multiply and simplify: x+8x+12 9 x+6 x+2
-
Use properties of logarithms to expand the logarithmic expression as much as possible. Assume a and b are positive real numbers. log 3 a
-
71 Show that the system is inconsistent. I x+y+z = 0 x + 3y+5z = 2 x + 2y + 3z = 8
-
Figure 10 shows a time-series graph for the death rate (deaths per 1,000 people) in the United States since 1900. (For example, the 1905 death rate of 16 means that, for each 1,000 people living at...
-
Could the owner of a business prepare a statement of financial position on 9 December or 23 June or today?
-
The following data are available for the thermodynamic properties of graphite and diamond: Assuming that the entropies and densities are approximately independent of temperature and pressure,...
-
Arrange these compounds in order of increasing acid strength: CH3COH .. HOC-COH HOCCH2CH2COH
-
Use the tables in this chapter to predict whether these equilibria favor the reactants or the products: CHH CH + CHCH-N-CHCH, NHCHC=C: + :NH c) CHC=C-H+ (CH3)3C-0: Dc0: CH3 CH3 + CHCH,CHCH, +...
-
Complete these equilibrium reaction sin the most reasonable manner possible using the curved arrow convention to show the movement of electrons in the reactions, Predict whether the reactants or the...
-
Suppose you observed that one-year T-bills are trading with a yield to maturity (YTM) of 4.75%. The yield spread between AAA and BB rated corporate bonds is 130 basis points. The maturity yield...
-
You have $36,000 to Invest in Sophie Shoes, a stock selling for $60 a share. The initial margin requirement is 65 percent. calculate your rates of return if the stock rises to $90 a share and if it...
-
New Flyer Industries has decided to expand its production of hybrid transit buses. The firm expects incremental cash flows of? $40 million per year for the next 10 years. The upfront cost of the...

Study smarter with the SolutionInn App


