Name the following alkenes, and tell which compound in each pair is more stable CH {a) H2C=CHCH2CH3
Question:
Name the following alkenes, and tell which compound in each pair is more stable
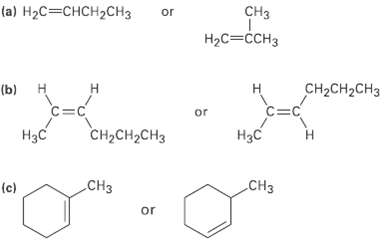
Transcribed Image Text:
CHз {a) H2C=CHCH2CH3 or Нас—ссHз CH2CH2CH3 Н (b) Н C=C or H3C CH-сH,CHз Нас CНз CHз (c) or
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a b c More stable H 1 CC H CH3 2Methylpropene disubstituted double bond H C...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which species in each pair is a better oxidizing agent under standard-state conditions? (a) Br2 or Au3+. (b) H2 or Ag+. (c) Cd2+ or Cr3+. (d) O2 in acidic media or O2 in basic media.
-
Which species in each pair is a better reducing agent under standard-state conditions? (a) Na or Li. (b) H2 or I2. (c) Fe2+ or Ag. (d) Br- or Co2+.
-
1. Which compound in each pair has a higher melting or boiling point? Explain. a. MgO or NaCI (melt) b. HBr or Kr (boil) c. CaBr, or CaCli(melt) d. SiH, or PH, (boil) 2. The molar heat of...
-
(a) By what percentage does your rest mass increase when you climb 30 m to the top of a ten-story building? Are you aware of this increase? Explain. (b) By how many grams does the mass of a 120-g...
-
In what way do effective teachers practice contingency leadership?
-
What is the meter reading, in ccf, indicated by each of the gas meters shown? a. b. c. d. 2 0 5 9 9 9 2 2 0 8 7 5 .3 2 9 7 9 2 2 0 2 2 7 7 9 23 0 1
-
Is a service a system?
-
Tetra Company's cost system assigns MSDA expenses to customers using a rate of 33% of sales revenue. The new controller has discovered that Tetra's customers differ greatly in their ordering patterns...
-
To discover whether an unknown gas is monatomic or diatomic, an experimenter takes a 2.0 L sample of the gas at STP and heats this sample to 100C at constant volume. (a) The gas absorbs 180 J of...
-
ExcelGo Bhd is a construction and property development company listed on the Main Board of Bursa Malaysia. The company operates in central area of Penang. The company closes its account on every 31...
-
Assign stereochemistry (E or Z) to the double bond in the following compound, and convert the drawing into a skeletal structure (red0):
-
Predict the products of the following reactions: (b) CH CHCCHH2CH (a) HCI H (d) CH CH2 (c) HBr CH-CH3CH2 H20 H2SO, (Addition of H20 occurs.)
-
Why is the contribution margin per unit of a critical resource more important than the contribution margin per unit of product in deciding which products to produce and sell?
-
Windsor, Inc. began operations on January 1, 2022. The following information is available for Windsor, Inc. on December 31, 2022. Accounts payable $4,600 Notes payable $6,440 Accounts receivable...
-
Blue Industries purchased a machine from Calico Corporation on October 1, 2016. In payment for the $144,000 purchase, Blue issued a one-year installment note to be paid in equal monthly payments at...
-
Bleakhouse Bakers produces breads and pastries. Bleakhouse is considering introducing a new breakfast item. They estimate materials costs at $4.00 per dozen and overhead costs of $2.02 per dozen....
-
Macy Pharmacy has a project which has the following cash flows. Year 0 -$200,000 Year 1 = $50,000 Year 2 $100,000 Year 3 $150,000 Year 4 $40,000 = Year 5 $25,000 = The cost of capital is 10%. What is...
-
For each of the following products and services, indicate whether it is more likely produced in a process operation or a job order operation. 1. Tennis balls 2. Tea 3. Solar panels 4. Maple syrup 5....
-
Why is air pressure highest at sea level?
-
Reichenbach Co., organized in 2018, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2018 and 2019. Instructions...
-
How much energy is released when 65.8 g of water freezes?
-
Draw the products of each of the following reactions indicated by the curved-arrow notation. (a) (b) CH CH NH2
-
Use the curved-arrow notation to indicate the flow of electrons in each of the transformations given in Fig. P3.33. (a) (b) (c) (d) (CHJ.NH-CH.-CH-C-OC-H5 BrC2 HC CH CH3 CH2 CH CH C: T:0:
-
Predict the products of each of the following reactions, and explain your reasoning. Use the curved-arrow notation to help you, and show the notation. (a) (b) AICI3 011 + H,N-CH-CH, -
-
Describe an experience when you communicated with someone that you thought were charismatic. What were some of the qualities that you were most impressed with? How did the individual influence you to...
-
On January 1, 2021, TulipCo sells equipment it manufactured to PoppyCo in exchange for a $1 million noninterest-bearing note due in two years. The note bears no explicit interest. It says only that...
-
Choose a planned learning activity for a group of children and select and prepare the resources required for the activity or explain and list all resource you will use. b) Explain in detail the...

Study smarter with the SolutionInn App


