Predict the products of the following reactions: (b) CH CHCCHH2CH (a) HCI H (d) CH CH2 (c)
Question:
Predict the products of the following reactions:
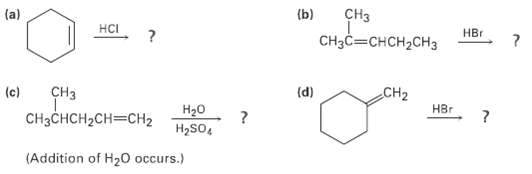
Transcribed Image Text:
(b) CHз CHзCCHсH2CHз (a) HCI Hвг (d) CHз CH2 (c) HBr CнзснсH-CH3CH2 H20 H2SO, (Addition of H20 occurs.)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 86% (15 reviews)
Strategy All of these reactions are electrophilic additions of HX to an alkene Use Markov...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant. H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e)...
-
Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
-
Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH CH2CH2CH2Br Hr SCH2CH3 (d) (c) Br2, ? H0z. 2 SH
-
(a) A proton is moving at a speed much slower than the speed of light. It has kinetic energy K1 and momentum P1. If the momentum of the proton is doubled, so P2 = 2p1 how is its new kinetic energy K2...
-
What relevance might contingency leadership have for dealing (a) men versus women, and (b) old versus young?
-
Bill Heckle's last electric bill is shown below. a. What was the previous reading? b. What is the present reading? c. How many kilowatt-hours of electricity did Bill use during the service period...
-
Is a product a system?
-
Rinehart Corporation purchased from its stockholders 5,000 shares of its own previously issued stock for $255,000. It later resold 2,000 shares for $54 per share, then 2,000 more shares for $49 per...
-
Mitchco produces it product through two processing departments: Cutting & Assembly. Information for the Cutting Department follows: DM CC Production % - Comp % - Comp Units Beginning WIP 3 0 , 0 0 0...
-
An auto manufacturer claims that the variance of the gas mileages in a certain vehicle model is 1.0. A random sample of 25 vehicles has a variance of 1.65. At = 0.05, is there enough evidence to...
-
Name the following alkenes, and tell which compound in each pair is more stable CH {a) H2C=CHCH2CH3 or H CH2CH2CH3 (b) C=C or H3C CH-H,CH C CH (c) or
-
What alkenes would you start with to prepare the following alkylhalides? CH-CH (a) (b) Br Br CI (c) (d) CH3CH2CHCH2CH2CH3
-
Given the following average compound annual inflation rates, how much would $1 be worth in terms of purchasing power five years from today as expressed in today's dollars? a. 5% b. 10% c. 15%
-
Forrest Company manufactures phone chargers and has a policy that ending inventory should equal 20% of the next month's budgeted unit sales. October's ending inventory equals 70.000 units. November...
-
3. Suppose the market is wild; it is modeled by . (a) What is the value of a Call? (b) What is the value of a Put? (c) Explain both answers in terms of finance.
-
Carla Vista Inc. has beginning work in process $30,300, direct materials used $283,200, direct labor $259.600, total manufacturing overhead $212,400, and ending work in process $37,000. What are the...
-
Consider the following information: Raw materials inventory, beginning $4,000 Raw materials inventory, ending 3,000. Net Purchases 25,000 Freight out 500% Decrease in Work in Process inventory Wip...
-
Sheridan Corp. had total variable costs of $224.200 total hied costs of $143500, and total revenues of $380.000. Compute the required sales in dollars to break even
-
About how much of solar radiation is intercepted by Earth? What portion of the solar energy intercepted by Earth is absorbed by the ground?
-
Rowland Textile Inc. manufactures two products: sweatshirts and T-shirts. The manufacturing process involves two activities: cutting and sewing. Expected overhead costs and cost drivers are as...
-
Methylamine has a vapor pressure of 344 torr at -25 C and a boiling point of -6.4 C. Find Hvap for methylamine.
-
In each of the following processes, give the products and classify each of the groups indicated by a colored label with one or more of the following terms: Bronsted base, Lewis base, Bronsted acid,...
-
The examples of incorrect curved-arrow notation in Fig. P3.36 were found in the notebooks of Barney Bottle brusher, a student who was known to have difficulty with organic chemistry. Explain what is...
-
Use the curved - arrow notation to derive three other resonance structures for anthracene. CC1..thmatummail three additional structures anthracene
-
Jeremy Pruitt Ltd is considering the replacement of a delivery truck. The current truck could last for three more years. Operating costs are 5000 per year. We are currently depreciating it at 4000...
-
As a long-term investment at the beginning of the 2024 fiscal year, Florists International purchased 30% of Nursery Supplies Incorporated's 8 million shares of capital stock for $30 million. The fair...
-
PJ Ltd Co. has been contracted to supply two products S & T worth 500 pieces, the joint cost function for the two varieties is given as: C = 100S2 +150T2. The quantity of S and T are not specified...

Study smarter with the SolutionInn App


