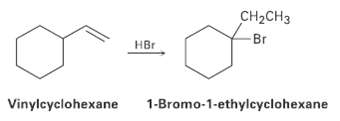
On treatment with HBr, vinylcyclohexane undergoes addition and rearrangement to yield 1-bromo-1-ethylcyclohcxane. Using curved arrows, propose a
Question:
On treatment with HBr, vinylcyclohexane undergoes addition and rearrangement to yield 1-bromo-1-ethylcyclohcxane. Using curved arrows, propose a mechanism to account for this result.
Transcribed Image Text:
CH-CHз Br HBr Vinylcyclohexane 1-Bromo-1-ethylcyclohexane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
Br 10 electrophilic add...View the full answer

Answered By

Ashish Jaiswal
I have completed B.Sc in mathematics and Master in Computer Science.
4.90+
20+ Reviews
39+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism to account for the reaction of benzene with 2, 2, 5, 5- tetra methyl tetra hydrofuran. H2SO4
-
Propose a mechanism to account for the following reaction: C CH2CI AICI3
-
Propose a mechanism to account for the formation of 3, 5-dimethylpyrazole from hydrazine and 2, 4-pcntancdionc. Look carefully to see what has happened to each carbonyl carbon in going from starting...
-
In what significant way do financial audits in government and not-for-profit organizations differ from those carried on in businesses? Your answer should also address the purpose of performance...
-
How would you characterize the leadership style of your favorite executive, athletic coach, or television character who plays a boss?
-
Mark works at two jobs. His primary job is a commission-paid job. Therefore, his monthly income from this job varies. His secondary job has a fixed monthly income. His quarterly dividend checks and...
-
Discuss the various benefits of integrating value and risk management processes.
-
Panarin Company entered into two contracts on the same date with Hjalmarsson Corporation. Panarin has provided the following analysis of price and cost for the contracts:...
-
Healthcare in the US is a mixture of capitalism and socialism. How does this impact healthcare market competition, mergers, and consolidations? What is the difference between the perceived and actual...
-
On March 1, 2018, Maxus Energy acquired a gold mine for $29,000,000. At the time of acquisition, the Proven and Probable gold reserves of the mine were estimated to be 470,000 ounces. During the...
-
What about the second step in the electrophilic addition of IIC1 to an alkenethe reaction of chloride ion with the carbocation intermediate? Is this step exergonic or endergonic? Does the transition...
-
Name the following alkenes, and convert each drawing into a skeletal structure: (b) (a)
-
Eurozone Slides Into Deflation for the First Time in Four Years Deflation occurred in 12 of the 19 eurozone countries in August. Analysts blamed the fall in part on lower oil prices and the delay of...
-
10% If the Bank of Canada purchases government securities worth $100, and assume that r (desired or required reserve ratio), in the above formula, is 10%, What will be the total expansion in money...
-
Rousan contributed $1900.00 at the beginning of every month to an RRSP. Interest on the account is 4.95% compounded monthly. If she converted the balance after 13 years into an RRIF paying 7.1%...
-
Badem Farm issued a 30 year, 6% semi-annual bond 5 years ago. The bond currently sells for $1,040. The company tax rate is 22%. What is the after tax cost of debt?
-
Sekersiz Industries has 60% debt in its capital structure. Its WACC is 8% and its cost of debt is 5%. The corporate tax rate is 20%. What is the company's cost of equity capita
-
Clair is a soccer player that just signed a 3-year contract with a new team. The contract will pay her as follows: $58,000 today $67,000 at the end of year 1 $73,000 at the end of year 2 $89,000 at...
-
What is wind chill?
-
Which of the following is NOT a magnetic dipole when viewed from far away? a) A permanent bar magnet. b) Several circular loops of wire closely stacked together with the same current running in each...
-
This table displays the vapor pressure of ammonia at several different temperatures. Use the data to determine the heat of vaporization and normal boiling point of ammonia. Temperature Pressure (K)...
-
Write a Bronsted acid-base reaction in which act as conjugate acid - base pairs. H0-H andCHOH/CH,:
-
In each of the following processes, complete the reaction using the curved arrow given; classify the process as a Bronsted acid-base reaction or a Lewis acid-base association/dissociation; and label...
-
In each of the following processes, complete the reaction using the curved arrow given; classify the process as a Bronsted acid-base reaction or a Lewis acid-base association/dissociation; and label...
-
An initially motionless test car is accelerated uniformly to 120 km/h in 7.98 s before striking a simulated deer. The car is in contact with the faux fawn for 0.515 s, after which the car is measured...
-
How do you define operational excellence? What factors are involved in achieving operational excellence? Who (within an organization) is responsible for operational excellence and why is this...
-
f(x) = x In(2x) Find f(x) for the function below....

Study smarter with the SolutionInn App


