Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
Question:
Predict the products of the following reactions:
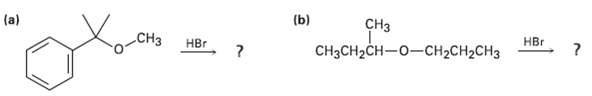
Transcribed Image Text:
(a) (b) CHз CH3CH2CH-0-CH2CH2CH3 CHз НЕr HBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a First notice the substitution pattern of the ether Bo...View the full answer

Answered By

Joram mutua
I am that writer who gives his best for my student/client. Anything i do, i give my best. I have tutored for the last five years and non of my student has ever failed, they all come back thanking me for the best grades. I have a degree in economics, but i have written academic papers for various disciplines due to top-notch research Skills.In additional, I am a professional copywriter and proofreader.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant. H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e)...
-
Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH CH2CH2CH2Br Hr SCH2CH3 (d) (c) Br2, ? H0z. 2 SH
-
Predict the products of the following reactions. When more than one product is expected, predict which will be the major product. (a) (b) (c) (d) OH H2SO4 heat H3PO4 heat BrNaocH, CH CH H,SO4 heat OH
-
Which is found in the java.sql package that come with the standard JDK? A. Only DerbyDriver B. Only MySqlDriver C. Only OracleDriver D. DerbyDriver, MySqlDriver, OracleDriver E. Only DerbyDriver and...
-
What are some of the issues with using financial metrics to evaluate IT?
-
The following data relate to the proportions in a population of drivers. A = Defensive driver training last year B = Accident in current year The probabilities are given in the accompanying Venn...
-
. A monopsonists demand for labor can be written as VMPE = 40 0.005ED. Labor is supplied to the firm according to w = 5 + 0.01ES. Thus, the firms marginal cost of hiring workers when it hires off of...
-
Find the following values using the equations and then a financial calculator. Compounding/discounting occurs annually. a. An initial $500 compounded for 1 year at 6% b. An initial $500 compounded...
-
Because of its high dielectric strength, SF6 (sulfur hexafluo-ride) gas is widely used as an insulator and a dielectric in HV applications such as HV transform-ers, switches, circuit breakers,...
-
TEK management attempts to maintain a reasonable ratio of debt to equity. Most firms prefer relatively low levels of debt in their capital structures. Why? What other approaches could TEK use to...
-
Rank the following halides in order of their reactivity in the Williamson synthesis: (a) Bromo ethane, 2-bromopropane, bromo benzene (b) Chloro ethane, bromo ethane, 1-iodopropene
-
Write the mechanism of the acid-catalyzed cleavage of tert-butyl cyclohexyl ether to yield Cyclohexanol and 2-methylpropene.
-
(Uniqueness of limit) show that if a sequence converges, its limit is unique
-
Two charged spheres are placed as shown above. The size of the sphere represents how much charge it has. Choose the position (indicated by A, B, C, or D) where there would be zero net force on an...
-
A coding supervisor performs an internal audit on the coding staff. The overall accuracy results are 85 percent and the expectation is 95 percent. She decides to eliminate the worst records from the...
-
. For the investment analysis calculations, you will need to know how to calculate before-tax cash flow for a three-year holding period, calculate equity reversion, and investment value. I would...
-
1. The stress-strain data of a compression test on concrete cylinders are plotted as below. Assume normal-weight concrete is used for the cylinders. The compressive strength is mostly near psi, the...
-
How can Sally improve her credit score. Her credit score is currently 668. She owes Belk $100 She owes Dicks Sporting Goods $50 She owes Lowes $25 She owes 5,000 on her 2022 BMW She made 25 late...
-
The ethical concern that Aor most likely raised is: A. KYC. B. suitability. C. confidentiality. Nomie Aor works for an international bank as a private wealth adviser. Aor speaks several regional...
-
Question 2 For an n x n matrix A = form) via (aij)
-
Redo Problem 4.11 using Aspen Plus. Problem 4.11 Steam is produced at 70 bar and some unknown temperature. A small amount of steam is bled off just before entering a turbine and goes through an...
-
Suggest a possible structure for a compound with the formula C7H12O that has the following IR spectrum and explain yourreasoning: 80 40 20 1718 cm 0- 500 1000 2000 1500 2500 3000 3500 4000 Wavenumber...
-
Suggest a possible structure for a compound with the formula C9H10O that has the following IR spectrum and explain yourreasoning: 100 - 50 1706 em- 4000 3500 3000 2500 2000 1000 500 1500 Wavenamber...
-
Forensic laboratories often have to identify various illicit drug samples. Explain how IR spectroscopy could be used to help distinguish between morphine and heroin. CH3CO, . NH NH CH,CO" ...
-
The veterinarian suspects a bleeding disorder in a young mixed breed dog. He requests that you perform a BMBT test on the dog. What other piece of information is necessary with any coagulation test?
-
.. Evaluate the following integrals: a) / b) c) d) x4 dx x-1 5x+1 dx (2x+1)(x-1) x3+4x+x-1 .3 x3 + x2 x+4x+3 x4 +5x +4 dx dx e) / 1 x3 - 1 dx
-
4. Evalute the following: (a) The region bounded by y = (3-2)' y = 0, x = 1, and x = 2 is revolved about the x-axis. (b) The region bounded by y = 1-x2, x = 0, x = 1,is revolved about the line x = 1....

Study smarter with the SolutionInn App


