Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry
Question:
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant.
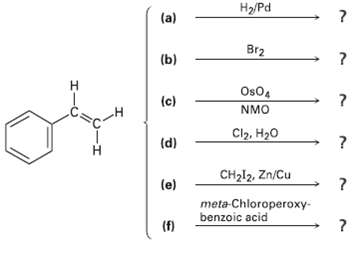
Transcribed Image Text:
H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e) meta-Chloroperoxy- benzoic acid (f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a b O c I CCH CCH HIC CCH HPd B...View the full answer

Answered By

Rehab Rahim
I am well versed in communicating and teaching in areas of all business subjects. I have helped many students in different ways from answering answers to writing their academic papers.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
-
Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH CH2CH2CH2Br Hr SCH2CH3 (d) (c) Br2, ? H0z. 2 SH
-
Predict the products of the following ozonolysis reactions. (a) (b) (c) (1) O2 (2) Me2S (1) 03 (2) Me S (1) O3 (2) Me S
-
Smart housing Inc. is negotiating a deal to build a house. The owner wants to start in early spring when the weather begins to moderate and build through the summer into the fall. The completion time...
-
High school students across the nation compete in a financial capability challenge each year by taking a National Financial Capability Challenge Exam. Students who score in the top 20 percent are...
-
The following data were collected from a standard 0.505-in.-diameter test specimen of a copper alloy (l0 = 2.0 in.): After fracture, the total length was 3.014 in., and the diameter was 0.374 in....
-
Use a stem-and-leaf plot to display the data shown in the table at the left, which represent the drunk driving cases registered at 30 strategic road intersections. Organize the data using the...
-
Below are seven factors that must be considered before finalizing a schedule. Explain how a base case schedule can change as a result of each of these: Introduction or acceptance of the product in...
-
We look at the detective novel market. Information gathered through a survey was used to Determine the following demand function and supply function: = 160-4 = 2-20 Graphically represent this console...
-
Phil Haddox and Russ French have decided to form a partnership. They have agreed that Haddox is to invest $120,000 and that French is to invest $180,000. Haddox is to devote full time to the...
-
From what alkene was the following 1, 2-diol made, and what method was used, epoxide hydrolysis or OsO4?
-
Suggest structures for alkenes that give the following reaction products. There may be more than one answer for some cases. H (b) CH CH3 /Pd CH2H2H2CH H2/Pd (d) CH Br CHH2CHCH (c) Br2 HCI ,...
-
Runway Apparel Inc. is considering two investment projects. The estimated net cash flows from each project are as follows: Each project requires an investment of $700,000. A rate of 15% has been...
-
Compare and contrast the strengths and weaknesses of two (2) federal mitigation programs. Be sure to explain how each functions to mitigate hazards in the United States.
-
At the end of the year 2022, Statistics South Africa estimated the total amount spent on consumption, government purchases, investment, and net export of goods and services to arrive at the country's...
-
According to the Google, Inc. Privacy Policy Litigation, on what date did Google announce their new universal privacy policy?
-
What would be considered "accumulated taxable income" for the year?
-
Explain how the reasons for the downward sloping of a demand curve for a normal good can be applied to the demand for healthcare goods and services.
-
What is the formula for the value of a forward contract in terms of the forward price?
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
One of these substances is a liquid at room temperature. Which one? a) CH 3 OH b) CF 4 c) SiH 4 d) CO 2
-
Ethyl bromide (0.IM) and HBr (0.1 M) are allowed to react in aqueous THE with 1 M sodium cyanide (Na+ -CN). What products are observed? Arc any products formed more rapidly than others? Explain.
-
What is the expected substitution product (including its stereochemical configuration) in the SN2 reaction of potassium iodide in acetone solvent with the following compound? (D = 2H = deuterium, an...
-
(a) Give the structure of the S*2 reaction product between ethyl iodide and potassium acetate. H,C-C potassium acetate
-
Korda International Inc. recently issued new securities (common shares and bonds) to finance a new project with a cost $16 million. The equity issued had a flotation cost of 8%, while the debt issued...
-
Khairul bought a television set with cash price of Rm6000. He paid a 10% down payment and the balance was settled by making a 24 monthly payments . If the interest was 8% per annum on the original...
-
Suppose a company produces a perpetual cash flow of $25 million per year and is expected to continue doing so in the infinite future. The company's capital structure currently consists entirely of...

Study smarter with the SolutionInn App


